Significant Digits
180 likes | 453 Views
Significant Digits. What are they? Where do they come from? Why do I care?. Significant Digits. The number of significant digits (figures) is a measure of the uncertainty of a measurement. The greater the number of significant digits, the more certain you are of your measurement

Significant Digits
E N D
Presentation Transcript
Significant Digits What are they? Where do they come from? Why do I care?
Significant Digits • The number of significant digits (figures) is a measure of the uncertainty of a measurement. • The greater the number of significant digits, the more certain you are of your measurement • The number of significant digits equals the number of digits that are certain, plus one additional digit , which is an uncertain digit.
Reading a Volumetric Device What is the correct measurement? Insert figure 3.13 Note the Meniscus
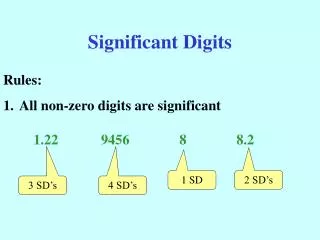
Determine the Number of Significant DigitsNon Zeros / Sandwiched Zeros • All non-zero digits are significant • Zeros “sandwiched” between two non-zero digits are significant
Determine the Number of Significant DigitsZeros with Decimal Point Present • If the measured quantity HAS a decimal point: • Start at the left of the number and move right until you reach the first non-zero digit. • Count that digit and every digit to its right as significant. • Decimal Present → Start at the “Pacific”
Determine the Number of Significant DigitsZeros Without Decimal Point Present • If the measured quantity DOES NOT HAVE a decimal point: • Start at the right of the number and move left until you reach the first non-zero digit. • Count that digit and every digit to its left as significant. • Decimal Absent → Start at the “Atlantic”
Determine the Number of Significant DigitsScientific Notation • Scientific notation can be used to show which zeros are significant: • Anything written in Scientific Notation is significant
Identifying Significant Digits Measurement • 0.0050 m • 0.0003056 L • 9.5 x 103 g • 56,980. cm3 • 56980 cm3 • 3.7890 x 104 s • 7000 g • 120500 mL • 9.5080 x 103 g Sig Figs
How many significant digits does each of the following numbers have? Measurement • 0.0050 m • 0.0003056 L • 9.5 x 103 g • 56,980. cm3 • 56980 cm3 • 3.7890 x 104 s • 9.50 x 103 g • 1.2 x 10-8 mL • 9.500 x 103 g Sig Figs • 2 • 4 • 2 • 5 • 4 • 5 • 3 • 2 • 4
Exact Numbers • Numbers that are a result of counting are exact • Definitions contain exact numbers. • For example 60 s in one min, 1000 mg = 1 g, 0.01m = 1 cm • Exact numbers do not limit the number of significant figures in an answer. • Exact numbers have the number of significant digits the calculation requires.
Operations with Significant FiguresMultiplying and Dividing • When numbers are multiplied and divided, the answer has the same number of significant digits as measurement with the least number of total significant digits. • 0.00569 x 0.91 = • 0.0052 • 9863 876.89 = • 11.25
Operations with Significant FiguresAddition and Subtraction • In addition and subtraction, the answer has the same number of decimal places as the measurement with the fewest number of decimal places:
Operations with Significant FiguresAddition and Subtraction • In addition and subtraction, the answer has the same number of decimal places as the measurement with the fewest number of decimal places: • 23.560 + 2.1 = • 25.7 • 0.0056 - 0.004067 = • 0.0015
Rounding • Answers need to be rounded to the correct number of significant figures • The round a number to the proper number of significant figures or decimal points, start at the right of the number and remove all digits needed to have the correct number of digits. • If the last digit removed is > 5, round up. • If the last digit removed is < 5 leave the remaining last digit the same.
Operations with Significant Digits • 0.00569 x 0.91 = • 9863 876.89 = • 3.26 / 125.4 = • 2.647 / 2.2×104 = • 23.560 + 2.1 = • 0.0056 - 0.004067 = • 324.54 – 25.6 = • 28.9 + 300.25 + 2.945 = • 10.07 – 3.1 =