Reaction Calculations
570 likes | 597 Views
This tool allows for the calculation and analysis of solution and reaction components in equilibrium phases, including gas phases, mixtures, and non-equilibrium reactions.

Reaction Calculations
E N D
Presentation Transcript
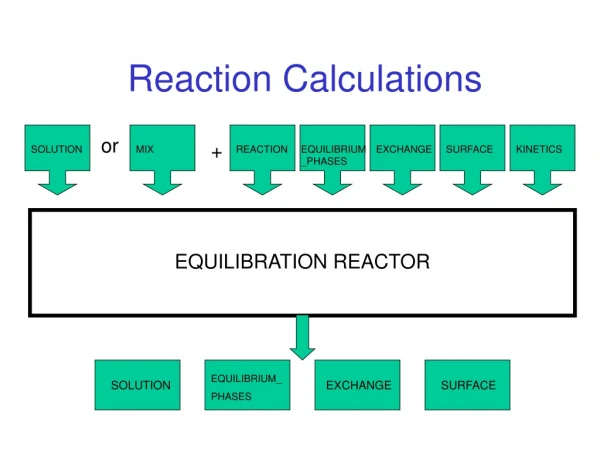
SOLUTION EXCHANGE SURFACE KINETICS MIX REACTION EQUILIBRIUM_PHASES SOLUTION EXCHANGE SURFACE EQUILIBRIUM_ PHASES Reaction Calculations or + EQUILIBRATION REACTOR
EQUILIBRIUM REACTIONS • SURFACE • EXCHANGE • SOLID_SOLUTIONS • EQUILIBRIUM_PHASES • GAS_PHASE • MIX
NON-EQUILIBRIUM REACTIONS • REACTION • REACTION_TEMPERATURE • KINETICS
CONCEPTUAL MODEL • Define initial solution (or mixture of initial solutions). 2. Define irreversible REACTION 3. Define EQUILIBRIUM_PHASES • Moles of each phase • SI or gas partial pressure
Reactions Implicit redox reactions
MIX: One or more SOLUTIONS • Type solution number • Type mixing fraction
R.1. Exercise Implicit Redox Reactionsmg/L • Define rainwater as SOLUTION 1 with log partial pressure of O2 = -0.7 and CO2 = -3.5. • Define END. • Define MIX by using solution 1 and mixing fraction 1. • Define END. • Run.
R.2. Questions 1. Explain the differences between the initial solution composition and the reaction (mixed) solution composition, particularly pH, pe, N(5), N(0), and N(-3).
Reactions Sequential reactions
SAVE and USE • Save results of calculations • Use previously defined SOLUTIONS, EQUILIBRIUM_PHASES, REACTIONs, etc • Use previously SAVEd SOLUTIONS, EQUILBRIUM_PHASES, etc
SAVE results from a “Reaction Calculation” Note: SOLUTION defines an “initial solution” calculation, which is automatically saved. Index numbers are used to keep track Index numbers do not need to be sequential
USE: Previously defined or SAVEd • USE includes KINETICS, MIX, REACTION, and REACTION_TEMPERATURE • Can USE previously SAVEd EQUILIBRIUM_PHASES, EXCHANGE, SOLID_SOLUTION, SOLUTION, or SURFACE
REACTION: Reactants and stoichiometry • Choose phase or type formula • Define relative stoichiometry
REACTION: Reaction amounts • Steps are a number of equal increments or • Steps are a specified list • Specify units
“Initial Solution” and Reaction CalculationsThe inscrutable END • Initial Solution/Speciation calculation—every SOLUTION • Reaction calculation—SOLUTION + reactant before END • Reactant may be any reactant keyword data block • USE can define solution or reactant • END—Do speciation and reaction • END—defines a “Simulation”
Speciation and Reaction CalculationsThe inscrutable END • Simulation 1 • Speciation calculation solution 1 • Speciation calculation solution 2 • Reaction calculation • SOLUTION 1 • EQUILIBRIUM_PHASES 2 • REACTION 3 • Simulation 2 • Speciation calculation solution 3 • Reaction calculation • SOLUTION 2 • EQUILIBRIUM_PHASES 1 • REACTION 3 • Simulation 3 • No speciation calculation • No reaction calculation—No Reactant defined
R.3. Exercise Sequential Reactions • Make an unsaturated zone water. Build on previous exercise with rainwater. Add SAVE solution 2 after MIX step and before END. After the END, add USE solution 2 and equilibrate (EQUILIBRIUM_PHASES) with CO2, log partial pressure –2, and calcite (SI = 0), save solution as solution 3. • Use solution 3, add 1 mmol CO2 (REACTION), equilibrate with calcite (EQUILIBRIUM_PHASES), save as solution 4.
R.4. Questions • What is the log pCO2 of the rainwater, rainwater, in redox equilibrium (the mixture), the mixture equilibrated with CO2 and calcite, and after reaction with CO2? • How many millimoles of calcite and CO2 reacted to make solution 3? • How many millimoles of calcite reacted to make solution 4 from solution 3?
R.5. Extra Credit Exercise • Use MIX and REACTION to evaporate rainwater 20 fold (at constant pCO2) before reaction with CO2 and calcite. Hint: You must remove water and water has 55.5 mol per kg.
Reactions Dedolomitization
Dedolomitization • Anhydrite dissolution • Calcite precipitation • Dolomite dissolution
R.6. Exercise • Make a ground water with log pCO2 = -2, equilibrium with calcite and dolomite. • React 50 mmol of anhydrite (CaSO4) in increments of 10 mmol. Maintain equilibrium with calcite and dolomite, allow anhydrite to precipitate if it becomes saturated.
R.7. Questions • What trends do you expect in water composition with anhydrite-driven dedolomitization? • Why is the following reaction misleading? CaSO4 + CaMg(CO3)2 = 2CaCO3 + Mg+2 + SO4-2 3. How does the water composition change from step 4 to step 5?
R.8. QuestionsPlummer and Sprinkle, 2001mg/L, S(-2) as H2S 1. Are these the trends of dedolomitization? 2. Anything else?
EQUILIBRIUM_PHASE:Dissolve Only • Force mineral not to precipitate
R.9. Extra Credit Exercise • Equilibrate seawater (major ions only) with calcite and dolomite. • Make a ground water with log pCO2 = -2, equilibrium with calcite and dolomite. • Mix ground water with seawater in fractions of .25, 0.5, .75. Maintain equilibrium with calcite; allow dolomite only to dissolve.
R.10. Questions • What reaction is calculated for seawater equilibration with calcite and dolomite? • What reactions are calculated for a carbonate reactions for the ground-water/seawater mixtures?
Reactions Organic decomposition
Organic Decomposition • Sequential removal of electron acceptors, usually in the sequence: • O2 • NO3- • MnO2 • Fe(OH)3 • SO4-2 • HCO3-
Redox Environments • Oxic—Dissolved O2 reduction CH2O + O2 = CO2 + H2O • Post-oxic—NO3-, MnO2, Fe(OH)3 reduction CH2O + 4Fe(OH)3 + 7CO2 = 4Fe+2 + 8HCO3- + 3H2O • Sulfidic—SO4-2 reduction 2CH2O + SO4-2 = 2HCO3- + H2S • Methanic—CH4 CH2O = CO2 + CH4
R.11. Exercise • Dilute seawater by 50 percent. • React diluted seawater with 50 mmol of CH2O in steps of 0.1, 0.2, 10, 20, 30, 40, and 50 mmol. Equilibrate with .1 moles of Fe(OH)3(a) and 0.0 mol of mackinawite. Print moles of reaction (-rxn) and total concentrations of O(0), C(4), C(-4), Fe(2), Fe(3), S(6), S(-2) (-totals) to the selected-output file (SELECTED_OUTPUT).
R.12. Questions • What sequence of electron acceptors is used? • Where is Fe(3) important? • Use Excel to plot the concentrations in the selected-output file (rxn is the x variable, set first line rxn from –99 to 0, omit d_mackinawite). 4. Why is C(4) not a straight line? 5. Why does Fe(2) increase after 30 mmol of CH2O is reacted. • Should a gas bubble form? • What trends are observed for sulfate reduction?
R.13. QuestionsPotomac Estuary Sediments • How is this similar to the sulfate reduction simulation results? • How does this differ from the simulation results?
R.14. Exercise • React pure water with 10 mmol of CH2O, maintain equilibrium with barite.
R.15. Questions • What is the barium concentration in mg/L? • Are there reactions other than sulfate reduction? • What other reactions could affect barium?
Sulfate ReductionNorman Landfill Barium concentration appears insignificant unless barium is lost to cation exchange or mineral precipitation.
R.16. Extra Credit Exercise • At pH 7.0, determine the pe indicated when concentrations are equal for each redox state of the following redox couples: As(5)/As(3), C(4)/C(-4), Fe(3)/Fe(2), N(5)/N(0), N(5)/N(-3), N(0)/N(-3), O2(/H2O*), S(6)/S(-2), U(6)/U(4). Hint: Use only SOLUTION and enter 1 umol/kgw of each valence state. *No entry is allowed in SOLUTION for H2O.
R.17. Questions • Iron is soluble as reduced ferrous iron, uranium is soluble as oxidized U(6). As organic carbon reacts, which would you observe first: (a) increase in iron, (b) decrease in uranium? • Thermodynamically, which nitrogen species to you expect to see in an oxygenated ground water? In a methanic water?
Cape Cod Sewage Plume • Iron reduction • No sulfate reduction Blue Plains, Washington D.C. Potomac River Sediment Pore Water • Iron reduction • Minor sulfate reduction • Intense methanogenesis
Reactions Sulfide oxidation
Sulfide Oxidation • Pyrite/Marcasite are most important reactants • Need Pyrite, Oxygen, Water, and bugs • Oxidation of pyrite and formation of ferric hydroxide complexes and minerals generates acidic conditions
R.18. Exercise • React the pure water with 10 mmol of pyrite, maintaining equilibrium with atmosphreric oxygen. • React the pure water with 10 mmol of mackinawite, maintaining equilibrium with atmosphreric oxygen. • React the pure water with 10 mmol of sphalerite, maintaining equilibrium with atmosphreric oxygen.
R.19. Questions • Write qualitative reactions that explain the pH of the 3 solutions. • What pH buffer starts to operate at pHs below 3? • Run the input file with wateq4f.dat database. What minerals may precipitate during pyrite oxidation?
R.20. Extra Credit Exercise • React the pure water with 20 mmol of pyrite, maintaining equilibrium with atmospheric oxygen and goethite. • Acid mine drainage is usually treated with limestone. Use the results of exercise 1 and equilibrate with O2, Fe(OH)3(a), and calcite.
R.21. Questions • Write a net reaction for the PHREEQC results for the low-pH simulation. • Looking at the results of the calcite-equilibrated simulation, what additional reactions should be considered?
Picher Oklahoma Abandoned Pb/Zn Minemg/L • Mines are suboxic • Carbonates are present • Iron oxidizes in stream
Reactions Aluminosilicate reactions
More stable Aluminosilicate Reactions • Disseminated calcite important in silicate terranes Olivine Pyroxene (augite) Amphibole (hornblende) Biotite mica • Bowen (Goldich) reaction series Ca-Feldspar Plagioclase Na-Feldspar K-Feldspar Muscovite Quartz