Functional Groups
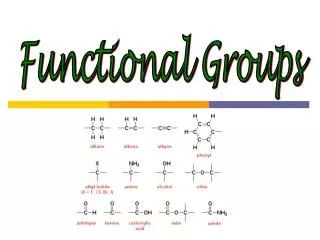
Functional Groups. Ethers. Ethers are much like alcohols except the oxygen is bound to two carbons instead of a carbon and a hydrogen. Unlike alcohols the ethers are not polar molecules so they will not dissolve in water

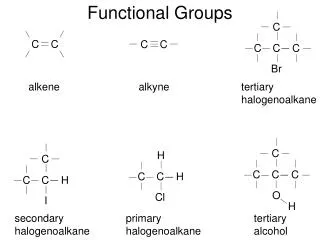
Functional Groups
E N D
Presentation Transcript
Ethers • Ethers are much like alcohols except the oxygen is bound to two carbons instead of a carbon and a hydrogen
Unlike alcohols the ethers are not polar molecules so they will not dissolve in water • Since they are not polar there is little interaction between molecules and so they have a much lower boiling point • DEE (-35°C)
Amines • Are compounds where a N is bound to 1, 2, or 3 saturated carbons. • Short amines have very strong odors
Once again amines are less polar than alcohols but more polar than ethers so their boiling points will be in the middle • Diethylamine is 55°C • Ethanol • Dimethyl ether
Carbonyl groups • The rest of the functional groups all have one common theme • A carbonyl group is a carbon that is double bonded to an oxygen
Ketones and aldehydes are very similar • Ketones are carbonyl groups attached to two carbons • Aldehydes are carbonyl groups attached to one carbon and one hydrogen
Carboxylic acid is where the carboxyl group is bound to a carbon and a hydroxyl group • They are called acids because the –OH group is very willing to donate a H to solution
More functional groups • Amides • Like amines these contain nitrogen • Difference: • They are connected to a carbonyl group
Esters contain a carboxyl group and are very similar to a carboxylic acid except the terminal hydrogen is replaced with a carbon or chain of carbons • Fairly short esters are found in a wide variety of odors and flavors
Polymers • A mer is the name given to a small molecular unit • A single mer is called a monomer • They can consist of 4-100 atoms • When monomers join together they form polymers
Where are they found? • Plastics – man-made polymers • Can be called synthetic polymers • Biopolymers • Plant cell walls • Proteins, DNA, complex carbohydrates
Synthetic Polymers • Are made with one of two reactions • Addition or Condensation
Addition Polymers • Addition polymers form from joining together two or more monomers • This reaction requires the presence of a double bond
In addition polymerization no atoms are gained or lost • Only electrons are shared between monomers
Polyethylene • There are two basic types of polyethylene • First is High Density Polyethylene (HDPE) • The second is Low Density Polyethylene (LDPE)
High Density • This polymer contains only straight chain polyethylene which makes it a dense and hard plastic
Low Density • Low density occurs when the chains branch. This leads to softer more flexible plastic.
Condensation Polymers • These polymers are formed by the joining of two or more monomers and their formation results in the loss of a small molecule. • This requires the use of functional groups • Can you think of any functional groups that contain oxygen or hydrogen?
Many different condensation polymers are formed from two separate compounds • Therefore they can be called copolymers • - two different monomers that work together to form a polymer
Condensation polymers can also be branched.