Periodic Table
200 likes | 270 Views
Explore the development of the periodic table, electron configurations, and the significance of atomic structure in chemistry. Understand how electron arrangements influence properties and behavior of elements. Learn about periodic trends in size and energetics.

Periodic Table
E N D
Presentation Transcript
Periodic Table Electron Configuration of the Atoms of the Elements
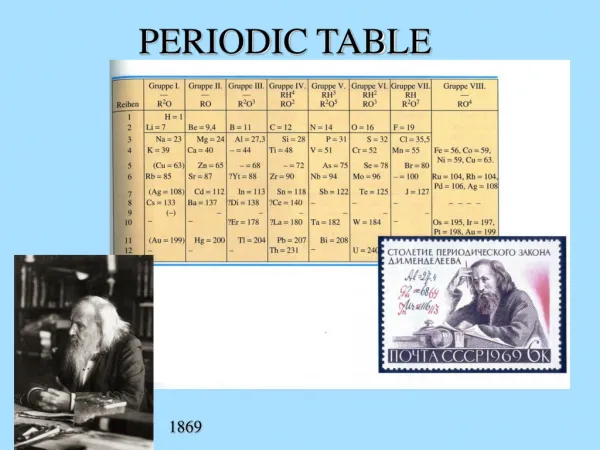
Periodic Table • Took form between 1868 and 1870 by Lothar Meyer in Germany and Dimitri Mendeleev in Russia. • Based on patterns in chemical formulae, chemical properties, and followed (generally) atomic mass. Atomic number was unknown concept at that time. • Atomic mass was finally sorted out after the Karlsruhe conference in 1860. These values were important in writing acceptable chemical formulae and were part of the organizational principles of the first periodic tables.
Contemporary Periodic Tables • Based on atomic number – the number of protons in the nucleus of an atom. • Nuclear structure of the atom discovered in 1911 by Ernest Rutherford. Mass of the atom is concentrated in a compact nucleus. • Antonius van der Broek hypothesized an atomic charge in the nucleus that was proven by Henry Moseley experimentally in 1913. The atomic charge (atomic number) is the number of protons in the nucleus and number of electrons of a neutral atom.
Atomic symbols and masses • Atomic symbols were set by international agreement (c.f., international federation of chemical societies in 1911 – IYC 2011 item.) • Atomic masses are fractions because elements exist as isotopes – same number of protons, different numbers of neutrons. The atomic mass tabulated is a statistical average over the contributions of all isotopes.
Electronic structure of atoms • The chemical properties of the atoms are determined by the organization of electrons in the atom, particularly those in the outer shell, the valence shell. • Chemically similar elements have the same, or nearly the same, valence shell electronic structures. • Electrons are organized into shells, subshells, and orbitals. Each orbital can have no more than 2 electrons.
Shell structure • Each electron shell has a limited number of subshells depending on which shell it is. Each subshell has a limited number of orbitals. • Shells are denoted by the n quantum number: 1, 2, 3, 4, … • Each shell n, has subshells numbered from 0,…,n-1. This provides the m quantum number. • Each subshell m has orbitals numbered from -m,…,0,…,m. This is the l quantum number. It represents the orientation of the orbital. • Each orbital has two electrons differing in spin: -1/2 or +1/2. This is the s quantum number.
Shell Structure Shell structure for the atoms in the periodic table appears thus – 1s 2s 2p 3s 3p 3d 4s 4p 4d 4f 5s 5p 5d 5f 6s 6p 6d 7s 7p 7d
Aufbau Principle • Shells and subshells are filled up in the following order: • 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f/5d 6p 7s 5f/6d 7p • In subshells with more than one orbital (p,d,f), electrons go in unpaired until each orbital has one electron and then they go in to make pairs.
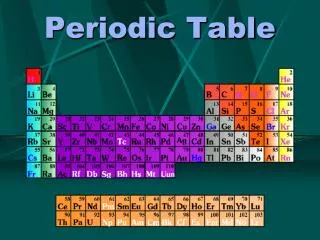
Electron configurations and chemical properties • Atoms with the same valence shell configurations behave similarly. They are called groups and listed vertically in the periodic table. • Rows in the period table are called periods and have changing properties from metal to non-metal ending up with the inert gases.
Valence shell structures • Gilbert Newton Lewis was the first to represent valence electron configurations in a paper published in 1916. • The maximum number of electrons in a valence shell is 8. No more than 8 dots are depicted. • These electrons may be lost to another atom or gained by another atom in ionic bonding. • These electrons may be shared with another atom in covalent bonding. • Atoms tend to achieve inert gas electron structures by forming ions or sharing electrons. • Atoms prefer to have half-filled or filled subshells if they cannot achieve inert gas structures (common for transition metals).
Periodic Trends -- Size • Within a group, the atomic radius increases with increasing atomic number. (related to increasing shell number). • Within a period, atomic radius decreases with increasing atomic number. The size depends on the average distance of its outermost electrons. There is no change in shell of the outermost electrons in a period. There is increasing atomic charge with increasing atomic number.
Periodic Trends -- Energetics • Within a group, the first ionization energy decreases with increasing atomic number. Size increases and the hold of the nucleus on outermost electrons drops. (There exceptions among the transition metals.) • Within a period, the general trend is an increase in ionization energy with increasing atomic number.
Example • Write the electron configurations for N, Ar, and Fe. How many unpaired electrons are in each?
Example • All of the lanthanides (rare earths) form stable compounds containing the 3+ cation. Of the few other ionic forms known, Ce forms the most stable 4+ series of ionic compounds and Eu the most stable 2+ series. Account for these ionic forms in terms of their electronic structures.
Example (3.103) • The most abundant isotope of lead is (a) How many protons, neutrons, and electrons are in this isotope? (b) What is the symbol for the lead isotope with 132 neutrons? (c) What is the atomic symbol and name of atom with the same mass number as in (b) and 131 neutrons?
Example (3.117) • Gallium has two naturally occurring isotopes: gallium – 69 (60.10%, 68.926 amu) and gallium – 71 (39.90%, 70.925 amu). What is the atomic mass of gallium?