Coactivators TAFs and the Mediators
570 likes | 779 Views
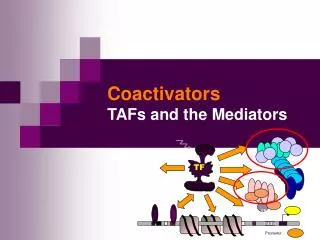
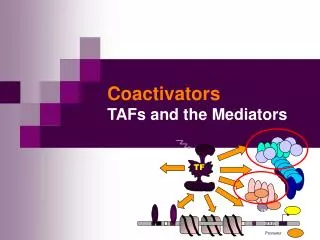
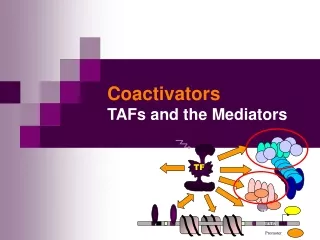
TF. TBP. TATA. Promoter. Coactivators TAFs and the Mediators. TBP. TFIIB. TFIIA. TFIIE. +. TFIIF. In vivo: In vitro:. TFIIH. basal trx.app. basal trx.app. OFF. ON. upstream transactivator. No activator response …. Something missing. ON.

Coactivators TAFs and the Mediators
E N D
Presentation Transcript
TF TBP TATA Promoter CoactivatorsTAFs and the Mediators
TBP TFIIB TFIIA TFIIE + TFIIF In vivo: In vitro: TFIIH basal trx.app. basal trx.app. OFF ON upstream transactivator No activator response …. Something missing ON Activation of basal transcription- the missing link? • RNAPII + GTF correct trx initiation in vitro, but do not respond to activators • Basal trx probably not occurring in vivo, eukaryotic promoters has to be activated by upstream trx factors • What is missing to reconstitute activator-dependent trx in vitro? • The coactivator was proposed to bridge the activator and other components necessary for transcription.
Activation of basal transcription • activator-dependent trx requires several additional actors: • basalt trx.apparatus - RNAPII + GTFs • Transactivators - sequence-specific DNA-binding transcription factors • Coactivators • Chromatin remodelling coactivator basalt trx.app. upstream transactivator Activators (ordinary TFs) don’t affect the basal trx.apparatus directly, but indirectly through coactivators and chromatin
The coactivator bridges Roeder, R.G. (2005) Transcriptional regulation and the role of diverse coactivators in animal cells. FEBS Lett, 579, 909-915.
”Bridge” Chromatin remodelling Coactivators = molecular bridges + chromatin remodeling TFs does not affect the basal transcriptional apparatus directly, but indirectly through coactivators coactivator basal trx.app. upstream transactivator
3 main types of general coactivators • 1. TAFs • TBP-associated factors (TFIID = TBP + TAFs) • Multiple complexes that contain TBP • Multiple complexes that contain TAFs • 2. Mediator/SRB-complex(holoenzyme components) • RNAPII- associated factors • 3. General cofactors • Non-associated factors
TF TBP TATA Promoter 1. TAFs as coactivators
1. Coactivators associatedwith TBP: TAFs • TAFs = “TBP associated factors” • TAFs - Tjians biochemical studies • Function in activator response • TFIID reconstituted from recombinant TAFs makes the basal transcription apparatus responsive to activators (def. coactivator) • Distinct TAFs for each transcription system • RNAPI: SL1 = TBP + TAFIs • RNAPII: TFIID = TBP + TAFIIs • RNAPIII: TFIIIB = TBP + TAFIIIs TAFs TBP
A280 Identification of coactivators - biochemical approach • Result: • Basal trx.activity requires: core RNAPII + GTFs • Activator-responsive trx. activity requires : core RNAPII + GTFs + Coactivator ?
Multiple TAFs with multiple activities • Large complex with 8 - 12 subunits • Ranging in size from 250 kDa to less than 20 kDa • Highly conserved proteins(Drosophila, humans, yeast) • Functions associated with subunits • hTAFII250 - HMG-box, bromodomains, serine kinase, binds the TAF-complex to TBP • dTAFII150 - binds INR + downstream (human: separate factor = CIF) • hTAFII135 /dTAFII110 - contacts Q-richTADs (absent in yeast) • hTAFII95/ dTAFII80 - WD40 repeat • hTAFII80 /dTAFII60 - histone H4 like - contacts acidic TADs • hTAFII55 - binds multiple activators, including P-richTADs • hTAFII31 /dTAFII40 - histone H3 like - contacts acidic TADs • hTAFII28 • hTAFII20 - histone H2B like • Structure • EM shows three to four major domains or lobes joined by narrower bridges, organized in a horseshoe-like structure around a central channel. Two configurations observed: open and closed
ConservedTAFs New nomenclature TAF1 = TAFII250 etc
basal trx.app. upstream transaktivator Specific functions of the TAF-complex • 1. main function: interaction with activators • Physical contact found between TAFs and specific activators • TAF-activator contact: each type of activator contacts a particular TAF • dTAF40 and 60 -- VP16, p53 (acidic TAD) • dTAF150 and 60 -- NTF-1 (Ile-rich TAD) • dTAF110 -- Sp1 (Q-rich TAD) • dTAF55 -- CTF (P-rich TAD) • Logic: a TF recruits TFIID to the promoter through specific TAD-TAF contacts and this stimulates PIC-assembly
synergy Tr.respons linear A B A+B Multiple contacts to activators synergy • Multiple TAF interactions might explain synergy • synergy = > additive (linear) transcriptional response • When two or more TFs together result in higher levels of activation than the sum of each factors individual contribution
Functions of the TAF-complex • 2. main function : TAFs bind core-promoter elements • TATA: through TBP • INR: dTAF150 specific interaction with the INR-motif • dTAF250 also implied • alternative anchoring of TFIID to PIC • TAFII250, together with TAFII150, mediates binding of TFIID to the Inr and can support Inr-mediated transcription. • +GTF-contact: TAF110 and TAF60 bind TFIIA and TFIIB TAFs basal trx.app. upstream transactivator
Functions of the TAF-complex • 2. main function : TAFs bind core-promoter elements • TATA: through TBP • INR: dTAF150 specific interaction with the INR-motif • alternative anchoring of TFIID to PIC • +GTF-contact: TAF110 and TAF60 bind TFIIA and TFIIB • DPE recognized through dTAF60 and dTAF40
TAFs with nucleosome structure? • Several subunits with histone-like elements • hTAFII80 /dTAFII60/ yTAFII60 - histone H4like • hTAFII31 /dTAFII40/ yTAFII17 - histone H3like • hTAFII20 /dTAFII30/ yTAFII68 - histone H2B like • In addition: hTAFII18 and hTAFII28 classfied as histone-like • Octamer-like structure possible?
Histone fold = dimerization motif • Histone fold frequently found in TAFs • More than half (9 out of 14) of the yTAFIIs contain a histone fold motif, and they specifically assemble into five histone-like pairs • The histone fold is the fundamental interaction motif involved in heterodimerization of the core histones, H4 and H3, and H2A and H2B.
TAFs with nucleosome structure? • 3. Main function - changing promoter topology or simply compact dimerization • Structuring element within the TAF complex? • Replacing nucleosomes, with DNA wrapped around - to mark active genes in mitosis?? • Counter argument - histones contact DNA through Args not conserved in TAFs • Probably simply to facilitate compact and tight protein–protein packing
The enzymatic functions of the TAF complex • 4+5+6. main function: enzymatic catalysis • 4. HAT-activity • histone acetyl transferase activity in TAFII250 • conserved activity in yeast, drosophila, humans mapped to central region • histone acetylation opens chromatin, important in gene activation (more later) • GTF substrates: TAFII250 acetylates TFIIE and TFIIF • In vivo substrates still open • Seminar: TAF1 activates transcription by phosphorylation of serine 33 in histone H2B • 5. Protein kinase • TAF250 has two kinase activities
TAFII250 is a bipartite kinase One Ser/Thr-kinase in the N-terminus (NTK) Another Ser/Thr-kinase in the C-terminus (CTK) In yeast: kinase domains in two separate proteins Substrates: see figure Itself - autophosphorylation GTFs, in particular TFIIF Kinase required in vivo Homologs TAFII130 and TAFII145 in yeast, TAFII230 and TAFII250 in Drosophila, TAFII250 and cell cycle gene 1 (CCG1) in mammals The versatile TAFII250
Recent novel functions: ubitiquination and binding acetylated histones • 6. Function: TAFII250 = a histone-specific ubiquitin-activating /conjugating enzyme (ubac). • TAFII250 mediates monoubiquitination of histone H1 • Monoubiquitination of histones has been correlated with activation of gene expression
Promoter recognition through TAFs bromo domains • 7. Function: Bromodomains • TAFII250 contains two tandem bromodomain modules that bind selectively to multiple acetylated histone H4 peptides. • Bromodomains may target TFIID to chromatin-packaged promoters
Summary of TAF functions 2. 6. 7. 5. 4. 1. 3. 2.
Summary of TAF functions (Drosophila) • Core promoter recognition factors • by binding to the Inr and DPE • by TBP:TATA box interactions, can orient TFIID on the DNA (single-sided arrows). • Certain TAFs also activator targets • capable of binding to activation domains in vitro (double-sided arrows). • Enzymytic activities • TAFII250 has two enzymatic activities, a kinase and an acetylase, that can modify proteins (squiggly arrows).
Sequential action • 1. Recruitment by bound activators
Sequential action • 2. Nucleosome and core promoter recognition and binding
Sequential action • 3. Chromatin dynamics
Sequential action • 4. Initiation and elongation of transcription
Hot debate on the importance of TFIID The TAF-complex in vivo: from general coactivator to gene-specific core-factor • TAF-coactivator-model under scrutiny • TAFs = biochemical artefacts or central actors in the activator response? • 1. interaction with activators - not verified in vivo • TAFs never found in genetic screens in yeast • Hypotheses on TAF function essentially based on in vitro studies (Tjian) • coactivator-model implies that most genes require the TFIID complex. • 2. interaction with core-promoter elements - supported by genome-wide analysis in yeast • Chimeric promoters Importance supported by in vivo evidence Only in vitro evidence Physiologically relevant? TAFs ! ? basalt tr.app. upstream transaktivator
The yeast attack - TAFs not universal factors required at all promoters • TAFs genes knocked-out - no global effects? • TAFs not universally acting • Each TAF controls only a subset of genes • Swap experiments suggest a role in core promoter recognition • The specificity of TAFs linked to recognition of core promoter
TFIID not the only TAF-complex- Multiple complexes contain TAFs • Presence of TAFII subunits not restricted to the well-known TFIID complex. Some TAFs have been found in other complexes, the function of which remains to be determined. • SAGA (yeast) • chromatin-remodeling complex that contains the histone-like yTAFII17, yTAFII60 and yTAFII68, and also yTAFII25 and yTAFII90. • STAGA (human) • Human version of SAGA • PCAF (human) • chromatin-remodeling complex with several histone-like TAFs • TFTC • TBP-free TAFII-containing complex • SAGA • chromatin-remodeling complex • Mot1 • Repressor that binds TBP-complex • NC2 • Global repressor that binds TBP (in absence of DNA) • Nots
Multiple complexes contain TAFs • Red • common to all • Dark blue • only in TFIID and TFTC, but not SAGA
TFIID not the only TAF-complex- Multiple complexes contain TAFs • Presence of TAFII subunits not restricted to the well-known TFIID complex. Some TAFs have been found in other complexes, the function of which remains to be determined. • SAGA (yeast) • chromatin-remodeling complex that contains the histone-like yTAFII17, yTAFII60 and yTAFII68, and also yTAFII25 and yTAFII90. • STAGA (human) • Human version of SAGA • PCAF (human) • chromatin-remodeling complex with several histone-like TAFs • TFTC • TBP-free TAFII-containing complex • SAGA • chromatin-remodeling complex • Mot1 • Repressor that binds TBP-complex • NC2 • Global repressor that binds TBP (in absence of DNA) • Nots
Multiple complexes with TBP 10x more TBP in a cell than there is of each of TAFs, SAGA, Mot1, NC2 and Nots
Many TBP-complexes - implications • TBP plays a role beyond TAFs • Trx probably regulered by several different TBP-containing complexes • TAF-complexes not global coactivators, but specific for subsets of genes • Unexpected importance of negative control of TBP? • Negative regulation of TBP so important that three different complexes (all essial for viability), have evolved - all bindning TBP.
TF TBP TATA Promoter 2. Mediator
3 main types of general coactivators • 1. TAFs • TBP-associated factors (TFIID = TBP + TAFs) • Multiple complexes that contain TBP • Multiple complexes that contain TAFs • 2. Mediator/SRB-complex(holoenzyme components) • RNAPII- associated factors • 3. General cofactors • Non-associated factors
Isolation of Mediator • Genetic screens (in yeast) for suppressors of truncations in the CTD of RNAPII • Supressors of cold-sensitive -CTD mutant • identified the SRBs (Suppressors of RNA polymerase B) components, which reside in a 1-2 Mda complex • Isolated biochemically (several systems) • activator-dependent in vitro assays • on the basis of its ability to stimulate activator-dependent trx in vitro • immunopurification assays based • activator affinity purification step • Based on physical interaction with various activators and the CTD of RNAPII • identified a variety of proteins, including Gal11, Srb proteins, Med proteins, and Rox3
The Mediator/SRB-complex is RNAPII-associated • Genetic isolation of supressors of CTD-deletion mutants SRBs • Biochemical isolation of a 20 polypeptide complex with coactivator properties • Consensus: Holoenzym = Mediator + RNAPII
The holoenzyme • Holoenzyme • a challenge to the linear assembly of PIC • A pre-assembled unit • Quantitative estimates argues against that all RNAPII is in the form of holoenzyme
Holoenzyme composed of several subcomplexes • Srb 2,4,5 and 6 subcomplex • Dominant suppressors • Srb 4+6 essential for most promoters • Srb 4 direct target for GAL4 • Srb7 • Essential, highly conserved from yeast to humans • Med-proteins • Associate with Srb 2-4-5-6 subcomplex through Srb4-Med6 contact • Repressor complex • several repressor-like proteins: Gal11, Sin4, Rgr1, Rox3 and Pgd/Hrs1/Med3 • Mutations of these components may derepress subsets of genes 10-fold • Kinase subcomplex (Srb10 CDK) • Recessive suppressors: Srb8, 9, 10 and 11 • Negative role • Srb10+11 forms a cyclin-CDK pair that phosphorylates Ser5 in the CTD tail • Srb10+11 can phosphorylate free RNAPII
Mammalian Mediator • Several coactivators for specific factors have turned out to be more general than first understood and are probably identical or variants of the Mediator-complex • TRAP- TR-associated proteins • Isolated as a coactivator for thyroid receptor (TR) • DRIP- vitamin D receptor-interacting proteins • Isolated as a coactivator for vitamin-D receptor (VDR) • Composition very similar to TRAP • ARC- activator-recruited cofactor • Isolated as a coactivator for SREBP-1a and Sp1, also coactivator for VP16, NFkB • Identical with DRIP • Human Mediator • Isolated as an E1A-interacting multicomplex with 30 polypeptides that bind activator-domains in E1A and VP16 • CRSP, NAT and SMCC • Contains several of the same subunits
Conservation and variability • Evolutionary conservation limited to a subset of mediator subunits • Probably different variant forms of Mediator
Functions of the Mediator/SRB-complex • Evidence for in vivo trx function of mediator • temp.sens. Mutation in SRB4: non-permissive temp all mRNA syntesis stops immediately • Mediator/SRBs like a control panel for trx • Kinase, activator like protein [ GAL11], proteins with repressor function (SIN4, RGR1) and other control proteins
Two variants of human Mediator- the smaller is the active version • Purification procedures identified two complexes • A larger 2 MDa complex termed ARC-L • Identical to complexes designated TRAP, DRIP, ARC, SMCC or NAT • Contains the cyclin-C–CDK8 pair (homologues of yeast Srb10+11) • A smaller 500-700 kDa complex termed PC2/CRSP • Lacks the cyclin-C–CDK8 pair • CRSP70 is present only in the CRSP complex • The larger complex appears to be transcriptionally inert, while the smaller CRSP complex is the active species on the promoter
The yeast mediator model of activator-dependent transcription • Different mediator proteins seem to have activator-specific roles • Activator contact • The three activators (GCN4, VP16 and GAL4) are shown binding to their DNA sites and recruiting yeast mediator to the promoter via a physical interaction with a mediator module
Different temporal orders of recruitment of mediator and RNAPII • 1. mediator RNAPII initiation of trx. • 2. Mediator + RNAPII trx initiated later • 3. RNAPII mediator initiation of trx • More complex than suggested by the holoenzyme model • Some evidence suggests that mediator functions in the reinitation step of the transcription cycle • a reinitiation intermediate/scaffold that contains TFIIA,TFIID, TFIIH, TFIIE, and mediator can be isolated • Re-entry of RNAPII as rate-limiting • The rate at which RNAPII gains access to the preformed ‘scaffold’ may become the rate-limiting step