Fixed Bed Reactor – 1
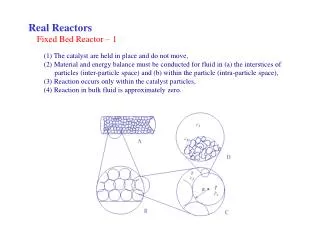
Real Reactors. Fixed Bed Reactor – 1 . (1) The catalyst are held in place and do not move, (2) Material and energy balance must be conducted for fluid in (a) the interstices of particles (inter-particle space) and (b) within the particle (intra-particle space),

Fixed Bed Reactor – 1
E N D
Presentation Transcript
Real Reactors Fixed Bed Reactor – 1 (1) The catalyst are held in place and do not move, (2) Material and energy balance must be conducted for fluid in (a) the interstices of particles (inter-particle space) and (b) within the particle (intra-particle space), (3) Reaction occurs only within the catalyst particles, (4) Reaction in bulk fluid is approximately zero.
Real Reactors Fixed Bed Reactor – 2 (5) Catalytic Reaction Steps (a) transport of reactants and energy from bulk liquid to the catalyst pellet surface, (b) transport of reactants and energy from pellet surface to pellet interior, (c) adsorption of reactants, chemical reaction and desorption of products at catalytic sites, (d) transport of products from the pellet interior to the surface, (e) transport of products into the bulk fluid. - usually one or at most two of the five steps are rate limiting and dictate, - most often it is the intra-particle transport step
Fixed Bed Reactors Catalyst Bed • Single pellet model is established by averaging the microscopic processes that occur within the intra-particle environment, • An effective diffusion coefficient is used to • represent the information about the • physical diffusion process • and pore structure, • A viable commercial catalyst must have sufficient • active sites to maintain a product formation rate • in the order of 1 mol/L h, • Catalyst pellets usually takes the shape of spheres • (0.3-0.7 cm), cylinders (0.3-1.3 cm O.D. and • L/O.D. = 3-4) and rings (ca. 2.5 cm)
Fixed Bed Reactors General Balances Catalyst Particle • Material Balance • where
Fixed Bed Reactors General Balances Catalyst Particle • Energy Balance • where
Fixed Bed Reactors Catalyst • Catalyst (usually metal sometimes also metal oxides) is often dispersed onto large surface area support material, • The support is often a refractor, metal oxide such as alumina. Silica, clay, zeolite, carbonaceous (e.g., activated carbon and graphite) are also popular support material. • The support often have surface areas between 0.05-100 m2/g.
Fixed Bed Reactors Catalyst Pellets – 1 • Catalyst pellets are made by tableting and extrusion methods. The latter is the more popular method, • Different pellet shape and size could be obtained by simply changing the extruder head, • The pellet shape and size could be optimized to increase mass transfer rates, while minimizing the pressure drop in the reactor.
Fixed Bed Reactors Catalyst Pellets – 2 • The pellet void fraction or porosity, where rp is the effective pellet density and Vg is the pore volume, • The pore volume range fro, 0.1-1 cm3/g pellet, • The pellet can possess either a uniform pore size or a bimodal pores of two different sizes, a large size to facilitate transport and a small size to contain the active catalyst sites.
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 1 • Material balance • Steady-state • Spherical coordinate system
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 2 • Boundary conditions absence of driving force
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 3 • Dimensionless equation - 1 • characteristic length: • dimensionless length: dimensionless concentration: concentration scale length scale
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 4 • Dimensionless equation – 2 • where
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 5 • Simplification • where
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 6 • General solution • Specific solution
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 7 • Concentration profile in pellet
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 8 • Total productivity in pellet • letting
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 9 • Effectiveness factor – 1 • where • = 1 : the entire pellet volume is reacting at the same high rate because reactant is able to diffuse quickly through the pellet, • = 0 : the pellet reacts at a slow rate, since the reactant is unable to penetrate into the pellet interior.
Single Pellet Reaction First-Order Reaction (1) Spherical Pellet – 9 • Effectiveness factor – 2
Single Pellet Reaction Example – 1 The first order, irreversible reaction took place in a 0.3 cm radius spherical catalyst pellet at T = 450 K. At 0.7 atm partial pressure of A, the pellet’s production rate is –2.5 x 10-5 mol/g-s, what is the production rate at the same temperature for a 0.15 cm radius catalyst pellet. Given:
Single Pellet Reaction Example – 2 • List the equations for (a) overall productivity, (b) effectiveness factor and (c) Thiele modulus for a first order reaction in a spherical pellet.
Single Pellet Reaction Example – 2 • Solve for Thiele modulus • where 2.125 mol/cm3–s (0.3 cm)2 = 0.007 cm2/s (1.9 x 10-5 mol/cm3) k (0.3 cm)2 = ( )0.5 0.007 cm2/s
Single Pellet Reaction Example – 2 • Solve for overall productivity of a smaller pellet 2.61/s (0.3 cm)2 = ( )0.5 0.007 cm2/s The smaller pellet has about 60 % better overall productivity! Note: this is only true when the system is within diffusion-limited regime!
Single Pellet Reaction First-Order Reaction Other Pellet Geometries – 1 • Governing equation
Single Pellet Reaction First-Order Reaction Other Pellet Geometries – 2 • Characteristic Lengths • Dimensionless equations
Single Pellet Reaction First-Order Reaction Other Pellet Geometries – 3 • Effectiveness factor – 1 • or
Single Pellet Reaction First-Order Reaction Other Pellet Geometries – 4 • Effectiveness factor – 2
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 5 • Positive reaction orders • Redefining Thiele Modulus
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 6 • Redefining the equations
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 7 • Effectiveness factor as a function of Thiele modulus n 1
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 8 • Effectiveness factor as a function of Thiele modulus n < 1
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 9 • Concentration profile within pellet with reaction order less than 1 n = 0
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 10 • Effectiveness factor can be approximated by the analytical solution for first order reaction n > 0 concentration profile effectiveness factor overall productivity
Single Pellet Reaction Other Reaction Orders Spherical Pellet – 10 • Effectiveness factor can be approximated by the analytical solution for first order reaction n > 0 concentration profile effectiveness factor overall productivity
Single Pellet Reaction Hougen-Watson - 1 Find the effectiveness factor for a slab catalyst geometry (1) Governing equation
Single Pellet Reaction Hougen-Watson - 2 (2) Transformation into dimensionless equation where (dimensionless adsorption constant)
Single Pellet Reaction Hougen-Watson - 3 (3) Effectiveness factor (4) Rescaling the Theile modulus
Single Pellet Reaction Hougen-Watson - 4 (5) Effectiveness factor versus Thiele modulus
Single Pellet Reaction External Mass Transfer - 1 Rapid EMT Slow EMT <
Single Pellet Reaction External Mass Transfer - 2 (1) The presence of external mass transfer resistance will only affect the boundary condition (2) Dimensionless boundary conditions x x
Single Pellet Reaction External Mass Transfer - 3 (3) Biot number (4) Dimensionless equation
Single Pellet Reaction External Mass Transfer - 4 (5) Solving the equation (6) Concentration profile in spherical pellet small B means large external mass transfer resistance large B means no external mass transfer resistance
Single Pellet Reaction External Mass Transfer - 5 (7) New definition of effectiveness factor (8) Effectiveness factor versus Thiele modulus for different Biot numbers small B means large external mass transfer resistance large B means no external mass transfer resistance
Single Pellet Reaction External Mass Transfer - 6 (9) Effects of external mass transfer resistance slope -1 slope -2
Single Pellet Reaction External Mass Transfer - 7 (10) Summary
Single Pellet Reaction External Mass Transfer - 8 (11) Observed versus intrinsic kinetic parameters - 1 Reaction-limited Diffusion-limited
Single Pellet Reaction External Mass Transfer - 9 (11) Observed versus intrinsic kinetic parameters - 2 Diffusion-limited Internal mass transfer-limited External mass transfer-limited
Catalyst Pellet General Balances (1) Material Balance where
Catalyst Pellet General Balances (2) Energy Balance where
Single Pellet Reaction Nonisothermal Condition - 1 (1) Material Balance (2) Energy Balance Practical catalyst pellet usually have high thermal conductivity and therefore heat transfer could often be neglected.
Single Pellet Reaction Nonisothermal Condition - 2 (3) Solving the two balance equations for constant properties therefore