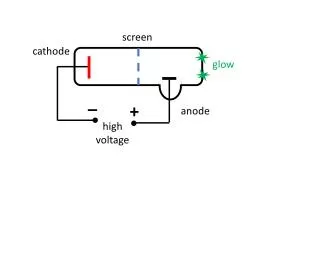
Electricity is transmitted over high voltage lines
Electricity is transmitted over high voltage lines. A Transformer: Converts a current at one voltage into a current in a separate circuit at a different voltage Entirely depends on the relative numbers of coils.

Electricity is transmitted over high voltage lines
E N D
Presentation Transcript
A Transformer: Converts a current at one voltage into a current in a separate circuit at a different voltage Entirely depends on the relative numbers of coils
A Transformer: Converts a current at one voltage into a current in a separate circuit at a different voltage Entirely depends on the relative numbers of coils
Remember….power is proportional to the square of the current: • P = I2R • The power lines have a small resistance, and so the power lost to heat will be P = I2R • Need to reduce the current to reduce power lost! • For the Transformer, V(in) x I(in) = V(out) x I(out) • If you go from 120 V to 120,000 V, your current is 1000 times less, and your power loss is a million times less!!
Structure of Helium Why isn’t the atomic mass 4 (actually 4.0028 amu)?
Structure of Helium Why isn’t the atomic mass 4 (actually 4.0028 amu)? Nuclear Binding Energy!
Why isn’t the atomic mass 4 (actually 4.0028 amu)? Nuclear Binding Energy! Mass of one neutron: 1.00866 amu Mass of one proton: 1.00728 amu 2 x neutron + 2 x proton = 4.0319 amu
Why isn’t the atomic mass 4 (actually 4.0028 amu)? Nuclear Binding Energy! Mass of one neutron: 1.00866 amu Mass of one proton: 1.00728 amu 2 x neutron + 2 x proton = 4.0319 amu SO….Nuclear Binding Energy = 4.0319 – 4.0028 = 0.029 amu Or, 4.82 x 10-29 kg But, E = mc2 = (4.82 x 10-29 kg)(3 x 108 m/s)2 = 4.3 x 10-12 J (Joining the neutrons and protons to make helium nucleus releases the energy)
Why isn’t the atomic mass 4 (actually 4.0028 amu)? Nuclear Binding Energy! Mass of one neutron: 1.00866 amu Mass of one proton: 1.00728 amu 2 x neutron + 2 x proton = 4.0319 amu SO….Nuclear Binding Energy = 4.0319 – 4.0028 = 0.029 amu Or, 4.82 x 10-29 kg But, E = mc2 = (4.82 x 10-29 kg)(3 x 108 m/s)2 = 4.3 x 10-12 J One gram of helium (by fusion) = burning 23 tons of coal
Radioactive Decay: Alpha Decay (atom loses helium nucleus) ( = Helium nucleus)
Radioactive Decay: Beta Decay (atom loses electron; neutron turns to proton) ( = Electron)
Radioactive Decay: Electron Capture (proton turns into neutron)
Zircon Crystals: Good for trapping in Uranium and Lead atoms (The oldest known zircon crystal in the solar system, from an Apollo 17 Moon rock: 4.42 billion years old)
Meteorites – oldest rocks on Earth Allende meteorite (carbonaceous chondrite)
Radioactive cascade The end result is a stable isotope (for U-238 Pb-206)
A Quick Look at Nuclear PowerU.S. Electricity Sector - Nuclear • nuclear power is major player in U.S. electricity industry • 19. 4 % of electricity • third major source behind: • coal: 46.6 % • natural gas: 21.5 % • characteristics: • despite no new plants since 1970s, percentage of electricity it produces has been growing • many plants being re-licensed for another 20-30 years • U.S. safety record has been stellar • no fatalities, no injuries
A Quick Look at Nuclear PowerGlobal Electricity Sector 61 new reactors (NEI, 2010) Taiwan – 2; Iran -1; Pakistan -1
Nuclear PhysicsNuclear Transformations • the nuclear structure of atoms is changed by three different mechanisms: • fission: splitting of heavy nuclei into two lighter ones with the releases of neutrons and energy • spontaneous • neutron-induced • fusion: combining of two nuclei to make a new, heavier nuclei • new nuclei has less mass than sum of two original nuclei • radioactive decay: spontaneous emission of either particle or electromagnetic radiation by nuclei • particle: alpha, beta, electron capture • electromagnetic: gamma • these processes are not influenced by physical conditions, e.g. pressure, temperature, etc.