Ionic Compounds: Bonds, Energy, and Properties
Learn about chemical bonds, electron structures, and properties of ionic compounds like lattice energy and crystal strength. Discover how metals interact with each other and form alloys. Understand the significance of electrolytes and the characteristics of covalent bonding.

Ionic Compounds: Bonds, Energy, and Properties
E N D
Presentation Transcript
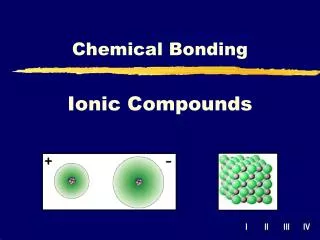
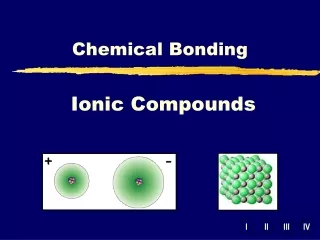
Ionic Compounds Chapter 8
Remember…. • Chemical bond • Electron-dot structure • Ionization energy • Electron affinity – how much attraction an atom has for electrons • Electronegativity • Octet rule • Cation • Anion
Atoms in contact will interact! • Based on electronegativity difference: • 1.8-3.3 ionic (metals with nonmetals) • 0.4-1.7 polar covalent (varying degrees) • 0.0-0.3 nonpolar covalent (2 nonmetals) • See page 169 • What about metals with other metals?
Brass White gold 14K gold Steel Cast iron Bronze Pewter Cu + Zn Au + Ni or Pd Au + Cu or Ag Fe + C Fe + C + Si Cu + Sn Sn + Cu or Sb or Pb Metallic atoms share their valence electrons freely in a “sea of electrons” to form alloys.
Pause for penny demo! http://www.slossfurnaces.com/
Ionic Crystalline arrangement (brittle/will shatter) High melting and boiling temperatures Ratio of atoms involved is determined by charges Non-conductive unless molten, dissolved in water Covalent Molecular arrangement Lower melting and boiling temperatures (may even be gases!) Ratio of atoms involved is determined experimentally Generally non-conductive Properties of other bonding:
Ionic Bond • Electrostatic force that holds oppositely charged particles together in an ionic compound • Binary ionic compounds – contain only two different elements • A metallic cation and a nonmetallic anion • Electrolyte – ionic compound whose aqueous solution conducts an electric current
Ionic Bond • # electrons lost must = # electrons gained • Calcium: 2+ charge • Fluorine: 1- charge • 1 Ca to every 2 F: CaF2
Example Ionic Bond • Sodium chloride • Na+1 , Cl-1 • Methods: (p. 216) • Electron configuration • Orbital notation • Electron-dot structures • Atomic models
Energy and Ionic Bonds • Endothermic – energy absorbed during a chemical reaction • Exothermic – energy released during a chemical reaction • Ionic compounds always exothermic reaction
Energy and Ionic Bonds • Lattice energy – energy required to separate one mole of ions of an ionic compound • Reflects strength of forces holding ions together • More negative lattice energy, stronger force of attraction
Crystal strength: • Determined by ionic radius • Smaller radii = higher lattice energy • Determined by ionic charge • Higher charge = higher lattice energy • KI < KF < LiF < MgO
Predicting ionic ratios • Based on charge ratios (“formula units” – simplest ratio of the ions) • Cations first, anions second • For example • Na 1+ and Cl 1- ; therefore, will combine 1:1 • NaCl “sodium chloride” • Na 1+ and S 2-; therefore, will combine 2:1 • Na2S “sodium sulfide” • Be 2+ and N 3-; therefore, will combine 3:2 • Be3N2 “beryllium nitride”
Oxidation Number • Charge of a monatomic ion (one-atom ion) • Also known as oxidation state • Group 1: +1 • Group 2: +2
D-block cations • Have varying oxidation numbers • Charges of these elements are indicated with Roman numerals (Stock method) • Cu (I) or Cu (II) • OR name changes (less common) • “-ic” means higher option (cupric = 2+) • “-ous” means lower option (cuprous = 1+)
Naming Binary Ionic Compounds • Name the cation (including charge if a d-block metal) and the anion with “-ide” • Sodium chloride Gold (III) iodide • Beryllium oxide Zinc nitride
Polyatomic ions • A group of atoms acting as one cation or anion • Memorize the chart on page 224 (Table 8.6) • Yes, all of it—test next Thursday • If more than one needed – parenthesis • Mg(ClO3)2 • Oxyanions- negatively charged polyatomic ion containing oxygen
Make another ‘A’ • Vocabulary • Memorize polyatomic ions • Read about alloys • Read about properties of ionic compounds • Practice writing formulas and names
Covalent bonding • …not ‘til next chapter! ;0) • The end!