Ionic Compounds
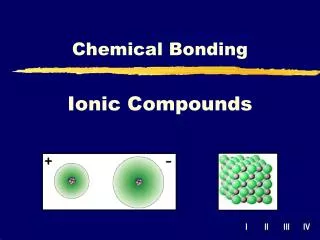
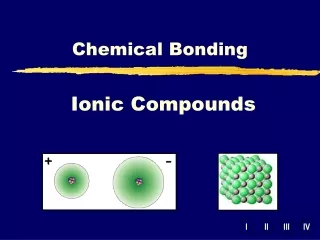
Ionic Compounds. Ionic Compounds. Ion:. Any atom that has a charge. Cat ion:. An ion with a + charge. an ion:. An ion with a - charge. Ionic Bonds. A bond that forms between (+) metals and (-) non metals. Electrons are transferred and then the opposite charges attract. Octet Rule.

Ionic Compounds
E N D
Presentation Transcript
Ionic Compounds Ion: Any atom that has a charge Cation: An ion with a + charge anion: An ion with a - charge
Ionic Bonds A bond that forms between (+) metals and (-) non metals. Electrons are transferred and then the opposite charges attract.
Octet Rule Element always move to become more stable. Most elements change their electron configuration to match the configuration of noble gasses. Elements want 8 valence electrons
1+ 2+ 3+ 3- 2- 1-
Rules for Naming Ionic Compounds 1. The first element (metal) name does not change. 2. The second elements name ending changes to -ide NaO MgCl2 Al2S3
Rules for Naming Ionic Compounds (except for polyatomic ions, use the name on the chart) SO42- = sulfate Na2SO4 = sodium sulfate
Rules for Naming Ionic Compounds 3. If it is a transition metal Roman Numerals must be used. FeCl2 FeCl FeCl3 The Roman numeral tells you the charge of the transition metal
Rules for Naming Ionic Compounds 4. There are no prefixes in ionic compounds
Rules for Writing Ionic Compounds 1.Charge must always cancel out + and – charges add up to zero. 2. Find the charges from the periodic table or the chart.
Rules for Writing Ionic Compounds 3.If the charges are the same they cancel out and it is a one to one ratio. Ca1O1 Ca2+ , O2-
Rules for Writing Ionic Compounds 4.If the charges are not the same then use the cross rule Ca2+Cl1- Ca1Cl2