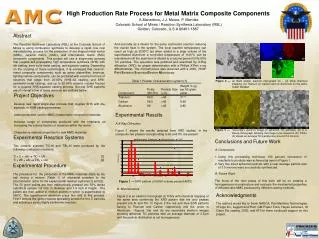
Development of Rapid Low-Cost Manufacturing for Metal Matrix Composites
The Reaction Synthesis Laboratory (RSL) at Colorado School of Mines is pioneering a combustion synthesis method that integrates self-propagating high-temperature synthesis (SHS) with die casting techniques. This novel approach aims to economically produce high-volume metal matrix composites (MMCs), ceramic matrix composites (CMCs), and intermetallic matrix composites (IMCs). The research focuses on maximizing ceramic volume fraction in composites for various applications, such as armor plates and vehicle components. Experimental results reveal a successful conversion of reactants to products and the synthesis of spherical TiC particles averaging 3.5 microns.

Development of Rapid Low-Cost Manufacturing for Metal Matrix Composites
E N D
Presentation Transcript
(b) (c) (a) (d) Figure 2 ― (a)Back scatter electronmicrograph (b) – (d) show chemical mapping (b) Titanium (c) Carbon and (d) Aluminum on the JOEL 7000F FESEM • Experimental Results • X-Ray Diffraction • Figure 1 shows the results obtained from XRD studies. In the composite two phases corresponding to Al, and TiC are present (a) (b) Figure 3 ― Secondary electron image of spherical TiC particles, (a) is a stereo-micrograph allowing the image to be viewed in 3D. Photo (b) shows an average TiC particle size around 3.5 microns Abstract The Reaction Synthesis Laboratory (RSL) at the Colorado School of Mines is using combustion synthesis to develop a rapid, low cost manufacturing process for the production of net shaped metal matrix (MMC), ceramic matrix (CMC), and intermetallic matrix (IMC) composite components. This project will use a single-step process that couples self propagating high temperature synthesis (SHS) with die casting, semi-solid metal casting, and squeeze casting. Depending on the coupling of these technologies, it is envisaged that ceramic-metal composite components, such as armor plates/tiles, bearings, fighting vehicle components, can be produced with volume fractions of ceramics that range from 20-60% (SHS-die casting, and SHS-semisolid metal casting), and up to 90-95% ceramic volume fraction for a coupled SHS-squeeze casting process. Several SHS systems are of interest a few of these systems are outlined below: High Production Rate Process for Metal Matrix Composite ComponentsA.Manerbino, J.J.Moore, P. Mendez Colorado School of Mines / Reaction Synthesis Laboratory (RSL)Golden, Colorado, U.S.A 80401-1887 And secondly as a diluent for the same exothermic reaction reducing the overall heat in the system. The local reaction temperature can reach as high as 2000ºC but when added to a large volume of the superheated Aluminum a controlled temperature of 1100°C can be maintained until the aluminum is diluted to a volume percent 20%-60% TiC particles. The specimen was polished and examined by X-Ray diffraction (XRD) for phase determination with a Philips X’Pert x-ray diffractometer. The microstructure was observed with a JOEL 7000F Field Emission Scanning Electron Microscope. Project Objectives Develop new rapid single-step process that couples SHS with die, squeeze, or SSM casting processes Lower production cost for MMC (metal matrix composite) materials Increase range of composites produced with the emphasis on increasing the volume fraction of ceramics within the matrix Characterize material properties for new MMC materials Experimental Reaction Systems Two ceramic systems TiC-Al and TiB2-Al were produced by the following combustion reactions: Ti + C + xAl TiC + xAl [1] Ti + 2B + xAl TiB2 + xAl [2] • Conclusions and Future Work • Conclusions • Using this processing technique 100 percent conversion of reactants to products was achieved as seen in Figure 1. • Very fine sized spherical particles with a near average particle size of 3.5 microns were successfully synthesized. (a) Experimental Procedure The procedure for the production of the MMC materials starts by dry ball mixing a mixture (Table 1) of elemental powders to the stiochiometric ratios for the experimental reaction systems [1] and [2]. The 50 gram pellets are then mechanically pressed into 55% dense cylindrical sample 1.0 inch in diameter and 1.0 inch in height. The pellets are then added to molten aluminum which is superheated to 960°C. The superheated aluminum plays two rolls in this process. First it acts as the ignition source spreading across the fine Ti particles and initiating a series highly exothermic reaction. B. Future Work The focus of the next phase of this work will be on creating a homogeneous microstructure and evaluate the mechanical properties of different ratio MMC produced by different casting methods. Figure 1 ― XRD pattern of 50/50 volume percent Al/TiC B. Microstructure Figure 2 is an electron micrograph at 1100x with chemical mapping of the same area confirming the XRD pattern that the only phases present are Al and TiC. In Figure 2 the red and blue EDS patterns belong to Titanium and Carbon respectively and the green is Aluminum. Figures 3(a) and (b) are secondary electron images showing spherical TiC particles with an average diameter of 3.5m and the particle distribution is not homogeneous. Acknowledgments The authors would like to thank NADCA, Pan Maritime Technologies, VForge Inc, Leggett and Platt, GM Power Train, Hayes Lemmerz, St Claire Die casting, DOD, and ATI for there continued support on this project.