The Periodic Table
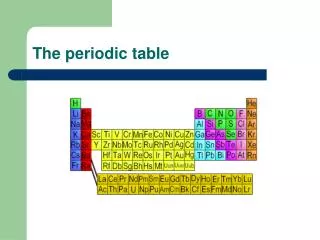
The Periodic Table. Here it is. Memorize it. Why is the Periodic Table important to me?. The periodic table is the most useful tool to a chemist. You get to use it on every test. It organizes lots of information about all the known elements. Pre-Periodic Table Chemistry …. …was a mess!!!

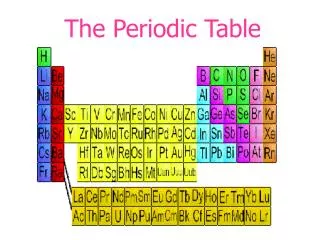
The Periodic Table
E N D
Presentation Transcript
Why is the Periodic Table important to me? • The periodic table is the most useful tool to a chemist. • You get to use it on every test. • It organizes lots of information about all the known elements.
Pre-Periodic Table Chemistry … • …was a mess!!! • No organization of elements. • Imagine going to a grocery store with no organization!! • Difficult to find information. • Chemistry didn’t make sense.
During the nineteenth century, chemists began to categorize the elements according to similarities in their physical and chemical properties. The end result of these studies was our modern periodic table.
In 1829, he classified some elements into groups of three, which he called triads.The elements in a triad had similar chemical properties and orderly physical properties. Johann Dobereiner (ex. Cl, Br, I and Ca, Sr, Ba) Model of triads 1780 - 1849
In 1863, he suggested that elements be arranged in “octaves” because he noticed (after arranging the elements in order of increasing atomic mass) that certain properties repeated every 8th element. John Newlands Law of Octaves 1838 - 1898
John Newlands Newlands' claim to see a repeating pattern was met with savage ridicule on its announcement. His classification of the elements, he was told, was as arbitrary as putting them in alphabetical order and his paper was rejected for publication by the Chemical Society. 1838 - 1898 Law of Octaves
His law of octaves failed beyond the element calcium. WHY? John Newlands Look at Be and Mg – they both are bordered on the right by the P BLOCK. How many electrons are s and p together? 1838 - 1898 Law of Octaves
In 1869 he published a table of the elements organized by increasing atomic mass. Dmitri Mendeleev 1834 - 1907
At the same time, he published his own table of the elements organized by increasing atomic mass. Lothar Meyer 1830 - 1895
Both Mendeleev and Meyer arranged the elements in order of increasing atomic mass. • Both left vacant spaces where unknown elements should fit. So why is Mendeleev called the “father of the modern periodic table” and not Meyer, or both?
Mendeleev... • stated that if the atomic weight of an element caused it to be placed in the wrong group, then the weight must be wrong. (He corrected the atomic masses of Be, In, and U) • was so confident in his table that he used it to predict the physical properties of three elements that were yet unknown.
After the discovery of these unknown elements between 1874 and 1885, and the fact that Mendeleev’s predictions for Sc, Ga, and Ge were amazingly close to the actual values, his table was generally accepted.
What to Do? • Follow the directions on the half sheet • Work alone or with a partner • Your table will have 7 columns and 6 rows • You MUST fill in missing observations on 5 elements (front table) • You MUST place the 9 unknowns and NAME EACH ONE! • You MUST designate AT LEAST 3 different physical/chemical properties DESIGNATED WITH A COLOR CODE
However, in spite of Mendeleev’s great achievement, problems arose when new elements were discovered and more accurate atomic weights determined. By looking at our modern periodic table, can you identify what problems might have caused chemists a headache? Ar and K Co and Ni Te and I Th and Pa
In 1913, through his work with X-rays, he determined the actual nuclear charge (atomic number) of the elements*. He rearranged the elements in order of increasing atomic number. Henry Moseley *“There is in the atom a fundamental quantity which increases by regular steps as we pass from each element to the next. This quantity can only be the charge on the central positive nucleus.” 1887 - 1915
Henry Moseley His research was halted when the British government sent him to serve as a foot soldier in WWI. He was killed in the fighting in Gallipoli by a sniper’s bullet, at the age of 28. Because of this loss, the British government later restricted its scientists to noncombatant duties during WWII.
After co-discovering 10 new elements, in 1944 he moved 14 elements out of the main body of the periodic table to their current location below the Lanthanide series. These became knownas the Actinide series. Glenn T. Seaborg 1912 - 1999
He is the only person to have an element named after him while still alive. Glenn T. Seaborg "This is the greatest honor ever bestowed upon me - even better, I think, thanwinning the Nobel Prize." 1912 - 1999
Atomic Symbols • Different Periodic Tables give different information. • All of them give Atomic Symbol (X). Usually they give the Atomic Number (Z). Some give more. • But even without all the fancy info, just knowing where you are on the table gives real, solid info! Let’s find out how!
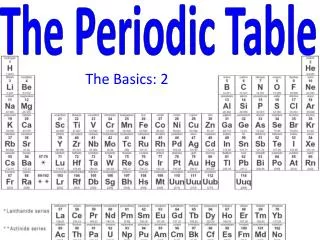
The horizontal rows of the periodic table are called PERIODS.
The elements in any group of the periodic table have similar physical and chemical properties! • Why?? • They have the same number of valence electrons. • They will form the same kinds of ions. The vertical columns of the periodic table are called GROUPS
Valence Electrons What Do We Want? WHY? s2p6
When elements are arranged in order of increasing atomic number, there is a periodic pattern in their physical and chemical properties. Periodic Law
Families on the Periodic Table • Columns are also grouped into families. • Families may be one column, or several columns put together. • Families have names rather than numbers. (Just like your family has a common last name.)
Hydrogen • Hydrogen belongs to a family of its own. • Hydrogen is a diatomic, reactive gas. • Hydrogen was involved in the explosion of the Hindenberg. • Hydrogen is promising as an alternative fuel source for automobiles
Metals • Metals are lustrous (shiny), malleable, ductile, and are good conductors of heat and electricity. • They are mostly solids at room temp. • What is one exception?
Group 1 Alkali Metals
Group 2 Alkaline Earth Metals
Groups 3-12 Transition Metals
Baby You’re A Rich Man • Why would rich folk take to using SILVER to make PLATES and UTENSILS? • Or Copper in their money? • It has been found that these metals have an ANTIBIOTIC quality; germs can’t stay alive on these metals. • Take note – what are door handles and stair rails usually made of? Why?
These elements are also called the rare-earth elements. They’re called: Lanthanide series Actinide Series InnerTransition Metals
Nonmetals • Nonmetals are the opposite. • They are dull, brittle, nonconductors (insulators). • Some are solid, but many are gases, and Bromine is a liquid.
Metalloids • Metalloids, aka semi-metals are just that. • They have characteristics of both metals and nonmetals. • They are shiny but brittle. • And they are semiconductors. • What is our most important semiconductor?
METALLOIDS ARE SHADED GREEN NON-METALS ARE SHADED BLUE