Fixed bed and fluidized bed
350 likes | 1.03k Views
Fixed bed and fluidized bed. Ref: BSL, McCabe & Smith. Why fixed (or fluidized) bed? Expensive Catalyst enzyme (immobilized) Large Surface area Used in reaction/adsorption/ elution (for example). Goal: Expression for pressure drop, try some examples. Fixed bed. Filled with particles

Fixed bed and fluidized bed
E N D
Presentation Transcript
Fixed bed and fluidized bed • Ref: BSL, McCabe & Smith • Why fixed (or fluidized) bed? • Expensive Catalyst • enzyme (immobilized) • Large Surface area • Used in reaction/adsorption/ elution (for example) • Goal: Expression for pressure drop, try some examples
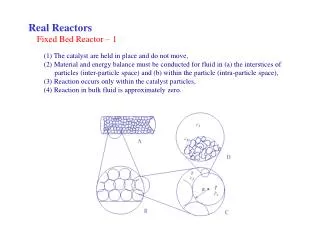
Fixed bed • Filled with particles • Usually not spherical • To increase surface area • To increase void fraction • To decrease pressure drop • For analytical calculation, assume all particles are identical • Usable, because final formula can be modified by a constant factor (determined by experiment)
Fixed bed • What are important parameters? • (For example, for adsorption of a protein from a broth) • rate of adsorption (faster is better) • saturation concentration (more is better) • From the product requirement (eg X kg per day), density and product concentration in broth ==> volumetric flow rate
Ap, Vp Fixed bed • Assume quick adsorption (rate of adsorption is high) • Calculate the surface area of particles needed for operation • Sphericity <=> specific surface area <=> average particle diameter • Sphericity • Volume of particle = Vp • Surface Area of particle = Ap • Surface Area of sphere of same volume (Vs =Vp) = As • Sphericity = As/Ap • May be around 0.3 for particles used in packed beds • lower sphericity ==> larger surface area As, Vs
Rings (Raschig,etc) Tarus saddle Pall Ring Fixed bed • Specific surface area • = Ap /Vp • Minimal value for sphere • Some books use S to denote area (instead of A) • Assume all the particles are identical • ==> all particles have exactly same specific surface area
Fixed bed • What is the pressure drop we need, to force the fluid through the column? • (i.e. what should be the pump spec) • We know the volumetric flow rate (from adsorption equations, productivity requirements etc) • We know the area per particle (we assume all particles are identical). And the total area for adsorption (or reaction in case of catalytic reactor). • Hence we can calculate how many particles are needed • Given a particle type (eg Raschig ring) , the approximate void fraction is also known (based on experimental results)
Fixed bed • What is void fraction? • Volume of reactor = VR • Number of particles = Np • Volume of one particle = Vp • Volume of all the particles = Vp * Np = VALL-PARTICLES • Knowing void fraction, we can find the reactor volume needed • Alternatively, if we know the reactor volume and void fraction and the Vp, we can find the number of particles
Fixed bed • To find void fraction experimentally • Prepare the adsorption column (or reactor....) and fill it with particles • Fill it with water • Drain and measure the quantity of water • (= void volume) • Calculate void fraction
Fixed bed • Since we know Vp, Np, e, we can find VR • Choose a diameter and calculate the length (i.e. Height) of the column (for now) • In normal usage, both the terms ‘height’ and ‘length’ may be used interchangeably (to mean the same thing) • Adsorption rate, equilibrium and other parameters will also influence the determination of height & diameter • To calculate the pressure drop • Note: columns with large dia and shorter length (height) will have lower pressure drop • What can be the disadvantage(s) of such design ? (tutorial)
Fixed bed • To calculate the pressure drop • You want to write it in terms of known quantities • Length of column, void fraction, diameter of particles, flow rate of fluid, viscosity and density • Obtain equations for two regimes separately (turbulent and laminar) • Consider laminar flow • Pressure drop increases with • velocity • viscosity • inversely proportional to radius • Actually, not all the reactor area is available for flow. Particles block most of the area. Flow path is not really like a simple tube • Hence, use hydraulic radius
Fixed bed - pressure drop calculation (Laminar flow) • To calculate the pressure drop, use Force balance • Resistance : due to Shear • Find Contact Area • Find shear stress • Until now, we haven’t said anything about laminar flow. So the above equations are valid for both laminar and turbulent flows
Fixed bed - pressure drop calculation (Laminar Flow) • Find contact area • To calculate the shear stress, FOR LAMINAR FLOW • Here V refers to velocity for flow in a tube • However, flow is through bed, NOT a simple tube
Fixed bed - pressure drop calculation (Laminar Flow) • Find effective diameter (i.e. Use Hydraulic radius), to substitute in the formula • Also relate the velocity between particles to some quantity we know • To find hydraulic radius ( and hence effective dia) • Hydraulic diameter
Fixed bed - pressure drop calculation (Laminar Flow) • Vavg is average velocity of fluid “in the bed”, between particles • Normally, volumetric flow rate is easier to find
Fixed bed - pressure drop calculation (Laminar Flow) • Can we relate volumetric flow rate to Vavg ? • Use a new term “Superficial velocity” (V0) • I.e. Velocity in an ‘empty’ column, that will provide the same volumetric flow rate • Can we relate average velocity and superficial velocity?
Fixed bed - pressure drop calculation (Laminar Flow) • Force balance: Substitute for t etc. • Volume of reactor (say, height of bed = L)
Fixed bed - pressure drop calculation (Laminar Flow) • Pressure drop • Specific surface area vs “average diameter” • Define “average Dia” of particle as • Some books (BSL) use Dp
In terms of specific surface area In terms of average particle diameter Fixed bed - pressure drop calculation (Laminar Flow) • Pressure drop • However, using hydraulic radius etc are only approximations • Experimental data shows, we need to multiply the pressure requirement by ~ 2 (exactly 100/48)
f Re • However Fixed bed - pressure drop calculation (Turbulent Flow) • Pressure drop and shear stress equations • Only the expression for shear stress changes • For high turbulence (high Re),
Hence, force balance • Volume of reactor (say, height of bed = L) Fixed bed - pressure drop calculation (Turbulent Flow) • We have already developed an expression for contact area
In terms of average particle diameter In terms of specific surface area Ergun Equation for packed bed Fixed bed - pressure drop calculation (Turbulent Flow) • Value of K based on experiments ~ 7/24 • What if turbulence is not high? • Use the combination of laminar + turbulent pressure drops: valid for all regimes!
Ergun Equation for packed bed Fixed bed - pressure drop calculation (Laminar OR Turbulent Flow) • If velocity is very low, turbulent part of pressure drop is negligible • If velocity is very high, laminar part is negligible • Some texts provide equation for friction factor
Fixed bed - pressure drop calculation (Laminar OR Turbulent Flow) • For pressure drop, we multiplied the laminar part by 2 (based on data) . For the turbulent part, the constant was based on data anyway. • Similarly...
Reynolds number for packed bed Fixed bed - pressure drop calculation (Laminar OR Turbulent Flow) • Multiply by 3 on both sides (why?) • Packed bed friction factor = 3 f Eqn in McCabe and Smith
Example • Adsorption of Cephalosporin (antibiotic) • Particles are made of anionic resin(perhaps resin coatings on ceramic particles) • void fraction 0.3, specific surface area = 50 m2/m3(assumed) • column dia 4 cm, length 1 m • feed concentration 2 mg/liter (not necessary to calculate pressure drop, but needed for finding out volume of reactor, which, in this case, is given). Superficial velocity about 2 m / hr • Viscosity = 0.002 Pa-s (assumed) • What is the pressure drop needed to operate this column?
Fixed Bed • What is the criteria for Laminar flow? • Modified Reynolds Number • Turbulent flow:- Inertial loss vs turbulent loss • Loss due to expansion and contraction • Packing uniformity • In theory, the bed has a uniform filling and a constant void fraction • Practically, near the walls, the void fraction is more • Ergun Eqn commonly used, however, other empirical correlations are also used • e.g. Chilton Colburn eqn 0.8 e 0.4 0.2 Edge Center Edge
Fixed Bed • Sphericity vs Void Fraction 1 f ~0.4 0 1 e
Fixed Bed • Alternate method to arrive at Ergun equation (or similar correlations) • Use Dimensional analysis
Fluidized bed • When the fluid (moving from bottom of the column to the top) velocity is increased, the particles begin to ‘move’ at (and above) a certain velocity. • At fluidization, • Weight of the particles == pressure drop (area) • Remember to include buoyancy
Fluidized bed: Operation • Empirical correlation for porosity • Types of fluidization: Aggregate fluidization vs Particulate fluidization • Larger particles, large density difference (rSOLID - rFLUID) ==> Aggregate fluidization (slugging, bubbles, etc) • ==> Typically gas fluidization • Even with liquids, lead particles tend to undergo aggregate fluidization • Archimedes number
Fluidized bed: Operation • Porosity increases • Bed height increases • Fluidization can be sustained until terminal velocity is reached • If the bed has a variety of particles (usually same material, but different sizes) • calculate the terminal velocity for the smallest particle • Range of operability = R • Minimum fluidization velocity = incipient velocity (min range) • Maximum fluidization velocity = terminal velocity (max range) • Other parameters may limit the actual range further • e.g. Column may not withstand the pressure, may not be tall enough etc • R = Vt/VOM • Theoretically R can range from 8.4 to 74
Fluidized bed: Operation 80 • Range of operation depends on Ar 40 R 0 100 104 108 Ar
Fluidized bed: Operation • Criteria for aggregate fluidization • Semi empirical • Particulate fluidization • Typically for low Ar numbers • More homogenous mixture