Bonding Considerations
Bonding Considerations. The following PowerPoint presentation is a self directed study of additional bonding considerations. It includes information above and beyond the ionic and covalent bonding that you have learned in class.

Bonding Considerations
E N D
Presentation Transcript
Bonding Considerations The following PowerPoint presentation is a self directed study of additional bonding considerations. It includes information above and beyond the ionic and covalent bonding that you have learned in class. You are expected to review and study this PowerPoint on you own time. When you feel you are ready, ask your teacher for a handout that will test your knowledge of these additional aspects of bonding nature.
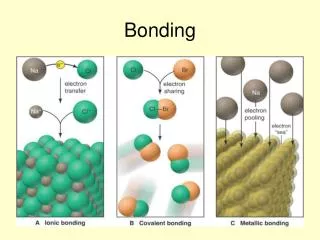
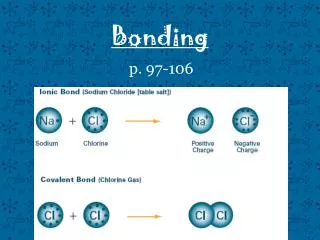
Let’s Review • IONIC BONDS exist when one atom transfers electron/s to another atom. Both atoms have attained a Noble Gas configuration. • COVALENT BONDS exist when two atoms share a pair of electrons between them in what is known as a SIGMA (s)bond. It is also possible for atoms to share more than one pair of electrons. These multiple bonds are known as PI (p)bonds. The atoms involved in bonding have attained a Noble Gas configuration.
VSEPR Theory Expanded • Consider the tetrahedral sp3 hybrid. It produces an atom with four bonding orbitals each separated by 109.5o. • However, this angle can be altered, in particular, by unshared electron pairs. Let’s consider two common examples of this phenomena.
Water • The bonding angle between the two Hydrogens in H2O with its two unshared electron pair lobes on the Oxygen is 104.5o. WHY? • The two unshared pairs, are not confined (restricted) between the nuclei of two atoms. • Therefore, they are able to expand to a greater degree and “squeeze” together the bonds of the two Hydrogen atoms.
Ammonia • The bonding angle between the three Hydrogens in NH3 with its unshared electron pair lobe is 107o. WHY? • The unshared pair on the Nitrogen is not confined (restricted) between the nuclei of two atoms. • Therefore, it is able to expand to a greater degree and “squeeze” together the bonds of the three Hydrogen atoms.
How can I see these angle changes for myself? • Using your Lewis structure directions, make a diagram of CH4 and NH3 and H2O. • Note the unshared pairs of electrons on the ammonia and water molecules. • Using the ball and stick sp3 models make models of methane, ammonia, and water. • Note that in the case of the ammonia and water there will be one and two holes respectively that aren’t filled. These are the unshared pairs. Fill them with gray sticks to represent the unshared electron pairs.
I’m having trouble visualizing these molecules. What can I do? • Methane: Note four sigma bonds with the H atoms which complete the octet of the Carbon.
I’m having trouble visualizing these molecules. What can I do? • Ammonia: Note the three sigma bonds with the H atoms. The lobe of unshared electrons are from the valence electrons of the N atom itself.
I’m having trouble visualizing these molecules. What can I do? • Water: Note the two sigma bonds with the H atoms. The two lobes of unshared electrons are from the valence electrons of the O atom itself. • Note the two lobes of unshared electrons on the lower right of the water molecule.
What have you learned so far? • QUESTIONS: • 1) What are all four angles for a tetrahedron? • 2) What is the angle between the two H’s in a water molecule? • 3) What are the angles between three H’s in an ammonia molecule? • 4) Why are the angles in water and ammonia less than a tetrahedron? ANSWERS: 1) 109.5o 2) 104.5o 3) 107o 4) The lobes of the unshared pairs expand, take up more space, and push the bonded atoms closer together.
COVALENT ~ Bonds within a single molecule IONIC ~ Bonds within a single crystal Bond strength is determined by BOND ENERGY, i.e. the amount of energy to break the bonds in Kilojoules per mole of bonds RANK ~ Strongest to Weakest Network Covalent Bonds Ionic Bonds Metallic Bonds Polar Covalent Bonds Non-polar Covalent Bonds Bond Strength ~ INTRAMOLECULAR BONDS
NETWORK COVALENT SOLIDS • Examples: C (diamond), SiO2 (silica sand) • Structural Particles: Atoms • Electronegativity Difference: Zero • Forces between Particles: Non-polar covalent bonds • Properties: Hard, very high-melting solids; nonconductors; insoluble in common solvents
Can I see a model of a diamond? • Here it is: C-C bonding! • Here’s SiO2 for you too! • SiO2 is sand and quartz
IONIC BONDS • Examples: NaCl (table salt), CaCO3 (calcite) • Structural Particles: ions (cation and anion) • Forces between Particles: Ionic bonds • Electronegativity Difference: >1.7 • Properties: High melting points; conductors in the molten state or water solution; usually soluble in water; insoluble in organic solvents
Ionic Crystals can have many different shapes. Table Salt Copper sulfate Triphylite
METALLIC BONDS • Examples: (sodium), Fe (iron), Au (gold) • Structural Particles: cations and mobile electrons • Forces between Particles: Metallic bonds • Properties: Variable melting points; conductors in solid state; insoluble in common solvents LEARN MORE ABOUT THE NATURE OF METALLIC BONDS BY CLICKING THIS LINK: www.ausetute.com.au/metallic.html
POLAR COVALENT BONDS • Examples: NH3 (ammonia), HCl (hydrochloric acid) • Structural Particles: Polar molecules • Forces between Particles: Polar covalent bonds • Electronegativity Differences: 0.2 – 1.7 • Properties: Generally higher melting points and boiling points than non-polar molecules; more likely to be water- soluble
NON-POLAR COVALENT BONDS • Examples: H2 (hydrogen gas), CCl4 (carbon tetrachloride) • Structural Particles: Non-polar molecules • Forces between Particles: Non-polar covalent bonds • Electronegativity Differences: 0 – 0.2 • Properties: Low melting and boiling points; often gas or liquid at 25oC; insoluble in water; soluble in inorganic solvents
What have you learned so far? • QUESTIONS: • 1) Intramolecular bonds are between what types of particles? • 2)What is bond energy? What unit is used? • 3) Rank the bond strength from highest to lowest. • 4) How are metallic bonds different from the other bonds? • ANSWERS: 1) Within a single molecule (covalent compounds) & within a single crystal (ionic bonds) 2) It is the energy to break a mole of bonds and the unit is kJ/mol. 3) Coordinate covalent, ionic, metallic, polar covalent, non-polar covalent 4) They involve a moving “sea of electrons.”
These are bonds between molecules (covalent compounds). Technically they are not bonds in the normal sense. Rather, they are attractive interactions. The bond strength is measured in Kilojoules per mole of bonds. Together these attractions are known collectively as van der Waals forces. RANK ~ Strongest to Weakest Hydrogen bonds Dipole – dipole interactions Dipole – induced dipole interactions Dispersion (London) forces Bond Strength ~ INTERMOLECULAR BONDS
HYDROGEN BONDS • A force exerted between an H atom bonded to an F, O or N atom on one molecule and an unshared electron pair on the F, O or N atom on another molecule. • The H on the molecule behaves almost like a bare proton because of the high electronegativities of the F (4.0), O (3.5) and N (3.0). • The small size of the H atom allows the unshared pair of the F, O or N to approach very closely. NOTE: This only happens with these three non-metals with their small atomic radii. • H bonding creates relatively high melting and boiling points compared to the low molar masses.
Water is affected by hydrogen bonding • The high surface tension of water is due to H bonds. • NOTE: H bonding creates higher melting and boiling points • Water, H2O,\; H bonds: • b.p. 100o C • molar mass 18 g/mol • Methane, CH4; no H bonds: • b.p. -161.6oC • molar mass 16 g/mol
DIPOLE-DIPOLE INTERACTIONS • Polar molecules experience an asymmetrical electronegativity difference higher than 0.2 across the molecule. • Such a molecule is called a dipole. • The dipoles line up as close as possible, positive end to negative end • There is an attractive force between adjacent molecules. This is known as the dipole moment. • Mr. Congdon’s Joke: What did the two dipoles say to each other? • You got a moment?
DIPOLE-INDUCED DIPOLE INTERACTIONS • A permanent dipole molecule such as Hydrogen fluoride (H-F) can “induce” (create) a temporary dipole moment in an adjoining non-polar molecule • The overall electron cloud of the molecule (or part of the molecule) will shift to create (+) and (-) poles • Note that these interactions are temporary and the non-polar molecule will shift its electron cloud position back to normal once the permanent dipole is no longer close d + d- Hydrogen fluoride
DISPERSION (London) FORCES • These forces involve attractions between temporary or induced dipoles in adjacent molecules. • At a given instant, the electron cloud around a non-polar molecule may shift from one side of the molecule to the other, thus inducing a dipole. • The temporary dipole induces a similar dipole in an adjacent non-polar molecule. • ALL molecules have dispersion forces. The strength of the forces depends on….. • the number of electrons that make up the molecule • The ease with which the electrons are dispersed to form temporary dipoles
London forces are responsible for holding much of your body together because of the many molecules involved. • Have you every split your lip or your knee bumping into something? OUCH! • You exerted enough bond energy to break the London forces in your lip or in your knee. • Remember London forces are the weakest of the intermolecular forces.
Once you have reviewed this PowerPoint, see your instructor for an evaluative problem set. 1) You will be asked to fill out several questions related to this PowerPoint. 2) You may refer back to the PowerPoint at any time. 3) The evaluative problem set will also give you book references. 4) You will also be asked to evaluate this PowerPoint and offer suggestions.