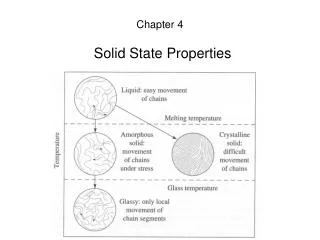
The Solid State
The Solid State. POINT DEFECTS RAJBENDRA CHOURASIYA PGT CHEM. K.V. GUNA (M.P.). 1. Vacancies. A vacancy is the absence of an atom in the lattice. In ionic crystals ( e.g NaCl ) vacancies occur in pairs (Na + Cl) so that charge balance is maintained. Also called a Schottky Defect.

The Solid State
E N D
Presentation Transcript
The Solid State POINT DEFECTS RAJBENDRA CHOURASIYA PGT CHEM. K.V. GUNA (M.P.)
1. Vacancies A vacancy is the absence of an atom in the lattice. In ionic crystals (e.gNaCl) vacancies occur in pairs (Na + Cl) so that charge balance is maintained. Also called a Schottky Defect. Vacancies allow diffusion through the crystal: Vacancy : pointdefect - associated with a point in the crystal
2. Interstitials Previously we discussed small tetrahedral and octahedral interstitial atoms within the close packed structure. If the interstitial atom is the same size as the close packed atoms, then considerable disruption to the structure occurs. Again, this is a point defect and requires much energy
3. Frenkel Defects Often a vacancy and interstitial occur together - an ion is displaces from its site into an interstitial position. This is a Frenkel Defect (common in e.g. AgCl) and charge balance is maintained. Frenkel defects can be induced by irradiation of a sample
METAL EXCESS DEFECT DUE TO ANION VACCANCIES THE ANIONIC SITES OCCUPIED BY UNPAIRED ELECTRONS ARE CALLED F- CENTRES. THEY IMPART COLOUR TO THE CRYSTALS WHEN THEY ABSORB ENERGY. NaCl – YELLOW LiCl – PINK KCl – VIOLET
METAL EXCESS DEFECT DUE TO PRESENCE OF EXTRA CATIONS THE EXCESS OF CATION MOVE TO INTERSTITIAL SITES AND THE ELECTRONS TO NEIGHBOURING INTERSTITIAL SITES. EXAMPLE - ZnO