E7: Soil
7.1 Discuss salinization, nutrient depletion and soil pollution as cause of soil degradation. E7: Soil. Where does it come from?. Physical means Weathering of rocks CaCO 3 + H 2 O + CO 2 -> Ca 2+ + 2HCO 3 - Chemical means

E7: Soil
E N D
Presentation Transcript
7.1 Discuss salinization, nutrient depletion and soil pollution as cause of soil degradation E7: Soil
Where does it come from? • Physical means • Weathering of rocks • CaCO3 + H2O + CO2 -> Ca2+ + 2HCO3- • Chemical means • Bacteria and fungi in the soil breakdown organic matter which then makes more soil
Components of soil • Inorganic matter • Largest component • From weathering of rocks • Contains • Silica • Silicates • aluminosilicates • Water/ Air • The amount affects how life forms survive and then decay • Organic matter • Bacteria and fungi convert organic matter into humus • A water insoluble mixture • Half it’s mass is carbon and approx. 5% nitrogen
Inorganic Matter • Silicates • Silicon is covalently bonded to four oxygens • Aluminosilicates • Al3+ has a similar size to silicon • Can replace the silicon to form aluminosilicates
Inorganic (cont’d) • If Al3+ replaces a Si4+ then in every four tetrahedrons there is 1 electron deficient • These ions + electronegative oxygen = ability to hold onto plant nutrients • Plant nutrients • K+ • Controls the amount of water that enters via osmosis • NH4+ • Synthesis of amino acids and proteins • Ca2+ • plant cell wall synthesis • Mg2+ • Synthesis of chlorophyll
7.1 Salinization • Accumulations of water soluble ions or salts in the soil due to continual irrigation to crops • How? • Small amounts of salts ( CaCl2, MgSO4, NaCl) are in water • When irrigation doesn’t wash them away they build up in the soil
salinity • Salinity is the total amount of dissolved salts in water; grams of salts per kilogram of water (g/kg) or as parts per thousand (ppt). • Seawater has 11 major constituents that make up more than 99.99% of all dissolved materials. • Although salinity may vary, the major constituents are well mixed and present in the same relative proportions.
Salts • Chloride (Cl-) 55.07% • Sodium (Na+) 30.62% • Sulfate (SO42-) 7.72% • Magnesium (Mg 2+) 3.68% • Calcium (Ca 2+) 1.17% • Potassium (K+) 1.10% • Bicarbonate (HCO3 -) 0.40% • Bromide (Br -) 0.19% • Strontium (Sr 2+) 0.02% • Boron (B 3+) 0.01% • Fluoride (F-) 0.01%
Salinity • Affects other properties of seawater, such as its density and the amount of dissolved oxygen.
Salinity • Significant Values • The average salinity of the world’s oceans is 35 ppt. • Freshwater has a salinity of <1 ppt. • Inshore waters with salinity values between 1 - 25 ppt are called brackish. • Waters with salinity greater than 40 ppt are called hypersaline.
Salinity • Addition of salts to pure water causes an increase in density. Salinity can be calculated by measuring the specific gravity of a water sample using a hydrometer, correcting for the effect of temperature and converting the readings to salinity by using conversion tables. Specific Gravity = density of sample Density of pure water
Salinity • Be sure hydrometer is clean • Fill 500 mL graduated cylinder with sample water • Determine the temperature of your sample • Place the hydrometer in cylinder and let settle. It should not touch the cylinder walls, and should be read from the bottomof the meniscus. • Read the specific gravity from the hydrometer scale • Using the specific gravity and temperature values, determine salinity from salinity table in Teacher’s Guide • Read three times. The values should be within 2 ppt of the average. Discard outliers.
Calibration • 35 ppt standard: • Measure out 17.5 g NaCl (table salt) and pour into a 500-mL graduated cylinder. • Fill the cylinder to the line with distilled water and carefully swirl the solution to mix the standard, until all salt crystals have dissolved. • Pour the solution into a 1-quart plastic bottle and label. • Prepare a blank using 500 ml of distilled water. • Follow the directions for a water sample. • Check technique every six months • .
How? • Bad irrigation • Small amounts of salts ( CaCl2, MgSO4, NaCl) are in water • When irrigation doesn’t wash them away they build up in the soil • Rise of the water table • Removal of native plants which leaves to reduction in evapo-transpiration • Soil water gets close to the surface and evaporates leaving behind the salts
Salinization Effects • Reduces plant growth • Increase in ions means a decrease in plants ability to take up water • Osmosis is affected • ---Suggested experiment: • effect of time, • temp., • size of seed, • type of soil, • water
Nutrient Depletion • Decreases soil quality = decline in crop yield • How? • crops that take nutrients and minerals from the soil and are continually harvested reduce soil quality
Countering Nutrient Depletion • Replenishing minerals and nutrients • Crop rotation • Nitrate/ Phosphate fertilizers • Chemicals end up in water supply • Leads to eutrophication (excess growth of aquatic life)
Soil Pollution or Contamination • Due to pesticides, herbicides, fungicides • Pesticides = kill insect pests • Lower crop yields – destroy flora/ fauna • Pollute ground water • Herbicides = kill unwanted plants or weeds • Fungicides = controls fungi
Soil Contamination • Nitrogen/ Phosphate fertilizers • Domestic/ industrial waste • Contain heavy metals (chromium, copper, mercury, lead, zinc) • Organic contaminants (PCBs, PAHs)
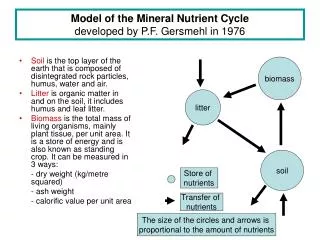
7.2 SOM • Soil organic matter • The mixture of non-living organic components present in the soil in various stages of decomposition • arising from thechemical • and biological action on plant matter and organisms
SOM • Amount of carbon in the soil is a measure of the organic matter present • Can be tested through titration using a dichromate(VI) solution
SOM (cont’d) • Contains • Humic substances • Contain phenolic acid (ArOH) and carboxylic acid (RCOOH) functional groups; weak acids • RCOOH RCOO- + H+ • The anion RCOO- binds to plant nutrients • Sugars • Amino acids • High molecular mass polysaccharides and proteins
Carboxylic acid • Anion: • Chelate to plant nutrients • Ca 2+ Fe 2+ Mg 2+ Ca 2+ • Zn 2+ Mn2+ Co2+ • Al 3+ >Mg 2+ >K • Higher the charge density, stronger the binding
polysaccharides • Commercial astragalus extracts have been standardized to 40-50% polysaccharides; • some sources claim ability to provide 70-90% polysaccharides.
Relevance of SOM in Preventing Soil Degredation • Plays important role in soil quality • Deserts (SOM= Sweet soil) • Organic matter + sand = improved plant growth • Holds onto water • Improves soil structure • Reduces soil erosion • E12.4 chemical function
SOM: Biological • Binds to nutrients – used by microbes and for plant growth • Bacteria and fungi decompose SOM • SOM Has large amounts of • Nitrogen 5% • Synthesis of proteins and chlorophyll • Phosphorus • Synthesis of enzymes and storage of energy • Sulfur • Synthesis of amino acids
SOM: Mineralization /ongoing process • Organic matter that contains nitrogen and phosphorus is broken down or mineralized to release nitrogen and phosphorus to the root • N, P, K, S
SOM: Physical • Allows stable forms of soil clusters • Reduces soil erosion • Increase water retention properties • Increases ability of air/ water to move through soil • More water affects thermal properties • Moist soil has a higher heat capacity than dry soil • Aka more water = smaller temperature change