BONDING
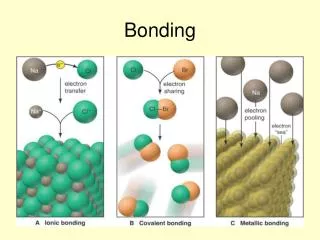
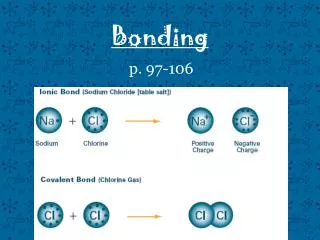
BONDING. REVIEW OF IONIC BONDING. Occurs when a metal transfers an electron to a nonmetal forming a charged particle . Intermolecular forces are Electrostatic Naming: Group IA,IIA,IIIA metals to nonmetals - Name the metal then the nonmetal with ‘ide’ ending.

BONDING
E N D
Presentation Transcript
REVIEW OF IONIC BONDING • Occurs when a metal transfers an electron to a nonmetal forming a charged particle. • Intermolecular forces are Electrostatic • Naming:Group IA,IIA,IIIA metals to nonmetals - Name the metal then the nonmetal with ‘ide’ ending. Transition metal to nonmetal - Name the transition metal, then its charge in Roman numerals in parentheses, followed by the nonmetal with ‘ide’ ending.
Metal to polyatomic ion – name the metal, then the polyatomic ion. If a transition metal, name the metal, its charge in Roman numerals, then the polyatomic ion. Why does this bonding occur? To achieve lower energy, that is, if the energy of the compound is lower than that of the separate atoms. Bond energy can be calculated using Coulomb’s Law: E = 2.31 x 10-19J nm Q1Q2 r If ‘E’ is negative, then the bonded pair has lower energy than the separate ions or atoms. Calculating for NaCl: E = 2.31 x 10-19K nm (+1)(-1) = -8.37 x 10-19J 0.276nm
Covalent Bonds Covalent bonds are formed when a nonmetalshares one or more pairs of electrons with another nonmetal. These are neutral molecules.
ELECTRONEGATIVITY: ELECTRONEGATIVITY IS THE ABILITY OF AN ATOM IN A MOLECULE TO ATTRACT ELECTRONS TO ITSELF. THERE ARE TABLES OF ELECTRONEGATIVITY VALUES FOR EACH ELEMENT. WHEN THE ELECTRONEGATIVITY DIFFERENCE BETWEEN TWO BONDED ATOMS IS 0.4 OR BELOW, THE MOLECULE IS CONSIDERED TO BE NON POLAR, COVALENT.
Polarity in bonds... Because different atoms have different electronegativities, one atom will pull more strongly on the electron pair in a bond.
Polarity... This uneven attraction for the electrons will cause the electron cloud, (the area where the electron is residing), to be more dense nearer to the atom with the higher electronegativity.
Polarity... The atom with the stronger attraction for the electrons gets a partial negative charge (because the negative electrons are closer to it). The other atom gets a partial positive charge.
SCALE OF ELECTRONEGATIVITY DIFFERENCES: 0---------0.4)------------1.6)--------------1.9------ NON POLAR POLAR PARTIALLY IONIC IONIC COVALENT COVALENT
Polar bonds forming non polar molecules... In a completely symmetrical molecule, the differences in electronegativity cancel each other out.
Example... 3.0 :Cl: .. I .. 3.0 :Cl : C : Cl:3.0 I 3.0:Cl: Cl = 3.0 C = 2.5
Forming of dipole attractions between polar molecules.... Because a polar molecule has a partial positive charge on one atom and a partial negative on another atom, there is a positive / negative attraction to other polar molecules.
The TWO POLES ( + and -) are referred to as a dipole. (aka dipole moment) These dipole -dipole attractions occur between polar molecules. The positive end of one attracts to the negative end of another.
dipole attractions... d+ d- d-d+ d+ d-
Hydrogen Bonding... Another important intermolecular force of attraction is Hydrogen bonding. This is a strong force of attraction between atoms of Hydrogen with either N, O, or F.
It is a stronger version of a dipole - dipole attraction. Nitrogen, Oxygen, and Fluorine are the most electronegative atoms and so the dipole is more pronounced in a molecule that contains H-N, H-O, or H-F
Because of these strong intermolecular attractions... Molecules which contain these bonds will be held together more tightly and thus be more difficult to separate. Molecules with H-bonds will have higher melting and boiling points, and viscosities than other molecules.
There are single, double, and triple covalent bonds. • A single bond occurs when one pair of electrons is shared between two atoms. • A double bond occurs when four electrons (two pairs) are shared between two atoms. continued...
continued... • A triple bond occurs where six electrons, (3 pairs) are shared between two atoms. Some elements do not form multiple bonds; (The Halogens, and some of the larger nonmetals).
Bonding order of some elements one single bond – the halogens, -F, -Cl, -Br, -I two bonds – oxygen, -O- , or = O three bonds – nitrogen, - N - , = N - , º N four bonds – carbon, - C - , - C = , = C = , - C º
The ‘how to’ of Lewis Dot Structures of Covalent Compounds 1. Count up the valence electrons on all of the atoms in the molecule. Total them up. 2. If there is a ‘+‘ charge, subtract the magnitude of the charge from the total.
Lewis Dot Structures continued... 3. If it has a ‘-’ charge, add the magnitude of the charge to the total. 4. Choose the element that there is less of to be the center of the molecule and place the others symmetrically around it.
Lewis Dot Structures continued... 5. Place dots (electrons) around each atom, two on each side, so that each atom has an octet. The two dots between two different atoms count toward the octet for each of those atoms. 6. Be sure to use no more or less electrons than the total from step 1.
Lewis Dot Structures continued... 7. If you run out of electrons before every atom has an octet, then you must move a pair from the outside of the atom adjacent to the lacking atom, to be shared between the two atoms to form a double (or triple) bond to satisfy the octet rule.
Example... CCl4 C = 4 valence electrons 4Cl = 7 x 4 valence electrons Total is 32 electrons to fill in. There is no charge. continued...
continued... C is the center of the molecule... :Cl : :Cl :C :Cl: :Cl: Each atom has an octet; 32 total e-
Example... SO4-2 S = 6 valence electrons 4, O = 6 x 4 valence electrons The total is 30 electrons plus two for the negative charge = 32 total e-
S will be the center... :O: nonbonding .. l.. electrons :O-- S –O: I :O: bonding electrons
Dot Structures of covalently bonded compounds with multiple bonds... Ex. CO2 C = 4 valence electrons 2, O = 6 x 2=12 valence total = 16 e- :O- C-O: THE ATOMS DO NOT HAVE COMPLETE OCTETS !!!
If two pairs of non bonding electrons are moved from the outside of the carbon to a position between C and O, octets will be satisfied.
Both of these are correct. The top one may be more stable than the bottom one. :O=C=O: or :O:::C :O:
Dot structures for ionic compounds show the non metal with a noble gas configuration (complete octet) and the metal with an empty outer shell. Na+ :Cl:
Resonance The last covalent example showed a structure that exhibited resonance. It appears that CO2 could exist it two different forms just by moving electrons around.
In a resonane structure the electrons are delocalized. This means that the electrons are shared by all of the atoms, not only the two that a simple diagram illustrates.
Other resonance structures... CO3 -2 NO3-1 Draw the dot structures of these ions and determine why they are resonance structures. Note: a single line can be used to represent an electron pair between two atoms, a double line - two pairs, triple - three pairs.
.. 24 e- 2- O O C O : : .. .. .. : : :: .. .. The double bond could appear between any one of the C-O bonds.
NO31- will have the same structure as CO32- NO31- 24 electrons 1- O = N - O Put in the non- l bonding O pairs yourself
Write all possible structures when drawing a resonance diagram. 1- O- N -O ll O O- N =O l O O=N -O l O Put brackets around the structures and place the charge outside the upper right corner.
Bond Lengths... Single bonds are longer than double bonds. Double bonds are longer than triple bonds. Triple bonds are the shortest and the strongest of the three. Double bonds are stronger than single bonds.
Bond Lengths In Resonance Structures... Experimentally, it was found that the bonds in a resonance structure are in between the lengths of single and double bonds.
Formal Charges... Why does NO31-have a -1 charge? Why does PO43- have a -3 charge? Why does NH3 have a 0 charge?
These charges can be determined by defining the formal charges on each individual atom in the particle.
how??? • Determine the valence on each atom in the compound. • Divide the shared electrons so that half of them count for each atom sharing them. • Nonbonding electrons count only on the atom that they belong to.
continued... • Determine the difference between the valence number of electrons and the electrons that were counted around the atom. That will be the charge on that individual atom.
Ex. NO31- O N O O .. .. : : :: .. .. .. : : ..
Counting electrons on each atom and then finding the difference from the valence... The left and bottom Oxygens have 7 electrons which are one more than their valences of 6. Therefore each has a charge of -1. The right Oxygen has 6 electrons which equals its valence, so it has a zerocharge.
Ex. continued... The Nitrogen has 4 electrons which is one less than its valence therefore it has a +1 charge.