Phase Changes
Phase Changes. Thermal Energy. Phase changes. A phase change is a reversible physical change when a substance changes from one state of matter to another. When ice melts to become water, it has undergone a phase change (and a physical change). The six common phase changes. Phase changes.

Phase Changes
E N D
Presentation Transcript
Phase Changes Thermal Energy
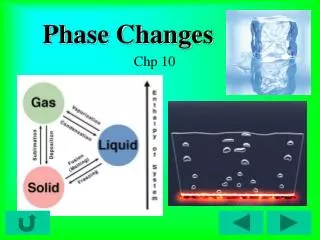
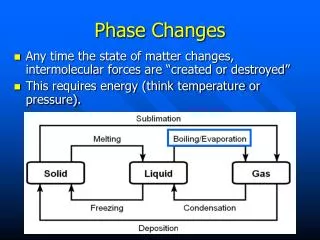
Phase changes • A phase change is a reversible physical change when a substance changes from one state of matter to another. • When ice melts to become water, it has undergone a phase change (and a physical change).
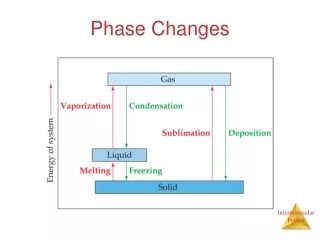
Phase changes Endothermic reactionsExothermic reactions (absorb energy) (release energy) Melting Freezing Solid Liquid Liquid Solid Vaporization Condensation Liquid Gas Gas Liquid Sublimation Deposition Solid GasGas Solid
Spotting a phase change on a diagram A phase change will always appear as a flat area on a temperature/ energy graph. A flat region shows that temperature remains constant while energy is converting matter from one state to another.
Energy required for a phase change The heat of vaporization is the amount of energy required for a liquid to change into a gas (vapor). Notice how much heat is required to change water into steam due to the length of the flat line. The heat of fusion is the amount of energy required for a solid to change into a liquid. The amounts of energy required for phase changes to occur is different in each substance. This graph shows a diagram for water.
Temperature and phase changes • One important point to understand about a phase change is that the temperature of the substance changing states of matter does not change until the phase change is complete. • So, if ice begins to melt to form water, the temperature of the melting ice and water will stay at the melting point until all of the ice has changed into water. This is true due to the large amount of energy required for matter to undergo a phase change.