Haemoglobin
Haemoglobin. By Maja & Jonathan. Protein Functions. Iron-containing oxygen-transporting metallprotein Essential for respiration Located in all red blood cells of vertebrates Around 35% of our blood (including water) Four iron atoms; can bind up to four oxygen molecules

Haemoglobin
E N D
Presentation Transcript
Haemoglobin By Maja & Jonathan
Protein Functions • Iron-containing oxygen-transporting metallprotein • Essential for respiration • Located in all red blood cells of vertebrates • Around 35% of our blood (including water) • Four iron atoms; can bind up to four oxygen molecules • Responsible for the red colour of blood (iron) • carries the oxygen from the lungs or gills to the cells
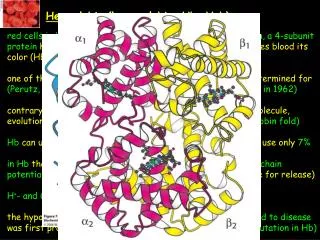
History • Many people did research for Heamogobin • Discovered in 1840 by Hünefeld • Description of revisable oxygenation by Felix Hoppe-Seyler • 1959 Max Perutz determined protein structure; resulted in Nobel prize for Chemistry in 1962 • Role in blood determined by Claude Bernard
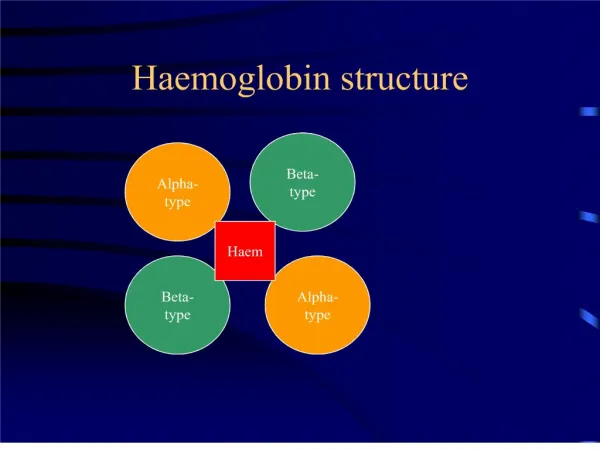
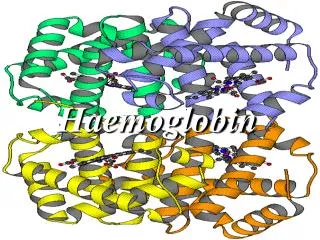
Primary Structure • The haemoglobin molecule consists of 4 polypeptide (globin) chains. In adults there are 2 alpha chains and 2 beta chains. • Peptide bond between each amino acidcalled residues
Secondary Structure • Joined amino acid residues coil to form sections of alpha helix, which stabilizes the structure • Structure fairly compact
TertiaryStructure • Mainbondinginvolvedin stabilising the structure of eachhaemoglobinchainis the attachment of a haemgroup (a porphyrin ring containing iron) --->the yellowball on the picture • No disulphidebridgesinvolved in the tertiarystructure of haemoglobin.
QuaternaryStructure • Twoalpha and two beta chainsareassociated to form a haemoglobinmolecule. • Morethan one peptidechain(multimericproteins). The manner in whichthesechainsfittogether (sort of like a puzzle) is the quaternarystructure.
Defectivefunction • Sicklecellanaemia • In the primarystructure: a single alteration in the aminoacidstructure- substitution of valineGTG (non polar) for glutamicacid GAG (polar) - only in the beta chain - causes the molecule to packdifferentlyinto the red bloodcells, causingsickling. • Thalassemia • inheritedcondition in whichreducedproduction of one of the globin chainsoccurs. The unpairedotherchainbuildsup in the cells, resulting in problems.
Works Cited • http://www.austincc.edu/emeyerth/tertiary.htm • http://faculty.stcc.edu/AandP/AP/AP2pages/Units18to20/blood/hemoglob.htm • http://www.biotopics.co.uk/as/haemoglobinproteinstructure.html