Element
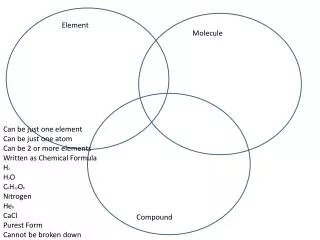
Element. An element is a substance composed of only 1 type of atom. All atoms of an element are alike. Compounds. Atoms of different elements join to form compounds . Atoms are held together by chemical bonds. Examples of Compounds. Water (H 2 0)

Element
E N D
Presentation Transcript
Element • An element is a substance composed of only 1 type of atom. • All atoms of an element are alike.
Compounds • Atoms of different elements join to form compounds. • Atoms are held together by chemical bonds.
Examples of Compounds • Water (H20) • Water has 2 hydrogen atoms and 1 oxygen atom • Table Salt (NaCl) • Rust (Iron Oxide) • Fe2O3
Chemical Properties • Chemical properties are the characteristics of a substance that describe what will happen when it interacts with other substances. • Ex: flammability, reactivity with oxygen, reactivity with acid
Physical properties – can be observed or measured without changing the composition of the matter Some properties of matter include hardness, texture, shape, temperature, flammability, size and color. Can matter change properties?
Matter can change properties. What are some examples of matter changing properties? www.wildwoodchapel.com/.../campfire.jpg www.learner.org/.../images/s4.ice_melt2.jpg
Physical changes alter the form of a substance, but not its identity. Example: When you boil away a pan of water, the water is still present as vapor. www.clipartguide.com/.../0060-0502-2515-5358.jpg
Chemical Changes • When a substance undergoes a chemical change, a new substance is formed. • The atoms are actually rearranged to form a new substance.
Signs of Chemical Change • Color change • Gas being produced • Temperature change • Precipitate formed • Properties changed
Examples of Chemical Change • Fireworks exploding • Baking a cake • Burning wood
Chemical Formulas • A chemical formula is a combination of symbols that represent the elements in a compound. • Example: Water H20
You cannot change the subscript – or you will have a different compound with different characteristics For example: CO2 is carbon dioxide, and CO is carbon monoxide. If you breathe too much carbon monoxide – you won’t wake up!
Chemical Equations A chemical equation is the way we write a chemical change using symbols. The arrow symbol (→ ) is read as “produces” or “yields” and means a chemical change takes place.
The elements or compounds written to the left of the arrow are the reactants, or what you start with. The elements or compounds written to the right of the arrow are the products, or what you finish with.
For example: C + O2 → CO2 Reads: “Carbon plus oxygen produces carbon dioxide”. What are the reactants? What is the product? Carbon and oxygen Carbon dioxide
Conservation of Mass • The amount of matter in a chemical reaction does not change. • The total mass of the reactants must equal the total mass of the products.
C + O2 → CO2 Count how many oxygen atoms are in the reactants. Count how many oxygen atoms are in the product. This is a balanced equation: demonstrating the Law of Conservation of Mass.
2H2O → 2H2 + O2 Read as “2 molecules of water produce 2 molecules of hydrogen and 1 molecule of oxygen.”
Balanced chemical equation • Has the same number of atoms of each element on both sides of the reaction.
Energy in Chemical Reactions • Every chemical reaction involves a change in energy. • Some reactions release energy and others absorb energy.
Endothermic vs. Exothermic Reactions • A reaction that releases energy in the form of heat is called exothermic. • Example: the reaction between gasoline and oxygen in a car engine; the engine gets hot enough to burn you if you touch it. • A reaction that absorbs energy is called endothermic. • Example: when baking soda reacts with vinegar, the reaction takes heat from the solution making it feel cool.