Element
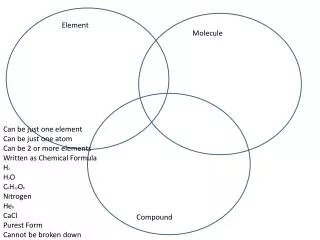
Elements and Compounds. Non-metals. Extraction of Sulphur. Element. Objective:. Compounds. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Extraction of Sulphur .

Element
E N D
Presentation Transcript
Elements and Compounds Non-metals Extraction of Sulphur Element Objective: Compounds A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Extraction of Sulphur. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
Elements and Compounds Non-metals Extraction of Sulphur Element Sulphur Compounds It is an abundant, multivalent non-metal. Under normal conditions, sulphuratoms form cyclic octatomic molecules with chemical formula S8. Elemental sulphuris a bright yellow crystalline solid at room temperature. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Sulphur is available in nature in both free and combined forms. Sulphur is obtained in combined form with certain metal ions viz copper pyrites, zinc blende, galena etc. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
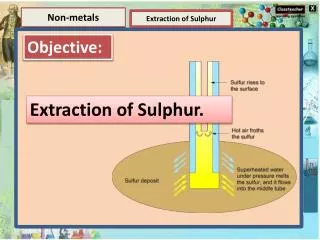
Elements and Compounds Non-metals Extraction of Sulphur Element Extraction of Sulphur Compounds A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. The Frasch process is a method to extract sulphurfrom underground deposits. It is the only economic method of recovering sulphurfrom elemental deposits. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.
Elements and Compounds Non-metals Extraction of Sulphur Element Extraction of Sulphur Compounds A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds. In the Frasch process, superheated water is pumped into the sulfur deposit; the sulfur melts and is extracted.
Elements and Compounds Non-metals Extraction of Sulphur Element Key points of Frasch Process Compounds Three concentric tubes are introduced into the sulphurdeposit. Superheated water (165 °C, 2.5-3 MPa) is injected into the deposit via the outermost tube. Sulphur(m.p. 115 °C) melts and flows into the middle tube. Hot air is introduced via the innermost tube to froth the sulfur, making it less dense, and pushing it to the surface. A compound is a substance composed of two or more elements, chemically combined with one another in a fixed proportion. Water, sugar, salt, carbon dioxide, methane etc are the examples of compounds.