Synaptic transmission
Synaptic transmission. Chapter 6 pages 156 - 169. Information transmission. Action potentials (APs) initiated by depolarizing stimulus Two general sources of depolarizing stimulus in neurons Receptor potentials from sensory transduction Information transmission between neurons

Synaptic transmission
E N D
Presentation Transcript
Synaptic transmission Chapter 6 pages 156 - 169
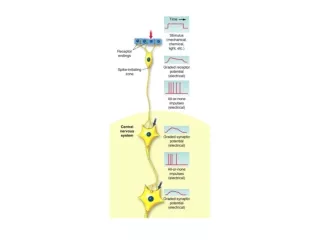
Information transmission • Action potentials (APs) initiated by depolarizing stimulus • Two general sources of depolarizing stimulus in neurons • Receptor potentials from sensory transduction • Information transmission between neurons • APs are intracellular events that encode information • Limited to individual cells by the cell membrane
Information transmission • Transmission of information usually requires multiple cells • Convergence - multiple cells with a single target • Divergence - single cell with multiple targets • Need mechanism to “inform” target cells of APs • Two types of mechanisms for transmitting information encoded by APs • Direct electrical coupling • Release of chemical messengers
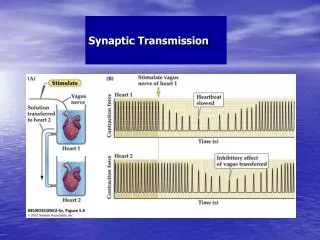
Electrical coupling • Cells are connected by ion channels that span two lipid bilayers • AP in one cell creates a voltage difference • Ions flow down voltage gradient and depolarize second cell • Advantages • Very rapid transmission of information • Disadvantages • Does not reflect all-or-none nature of APs (any depolarization is transmitted) • Effects on target cell limited to depolarization or hyperpolarization
Electrical coupling • Used to coordinate contraction in cardiac and smooth muscle • Coordinated contraction of heart to optimize blood flow • Coordinated contraction of smooth muscle lining digestive system and other organs • Recently found to have important role in some areas of the brain • Synchronizes oscillatory activity in small networks of interneurons • May be important for timing or gating of information transmission
Chemical coupling • AP can lead to release of chemical messenger • Hormones • Neurotransmitters • Advantages • Ligand release coupled to APs • Can evoke a variety of responses in target cell • One way communication • Disadvantages • Slower than electrical coupling • Juxtaposition of chemical release and target receptors reduces transmission delay • By far most common method of information transmission in body • Neuron → neuron • Neuron → muscle • Neuron → gland or organ
Which of the following is an advantage of electrical coupling? • Signal initiated only in response to APs • Faster than chemical coupling • Produces a variety of postsynaptic effects • Maintains one way communication • All of the above
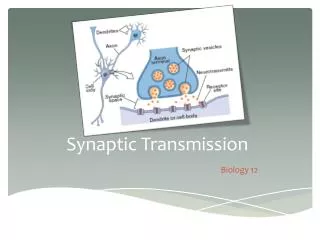
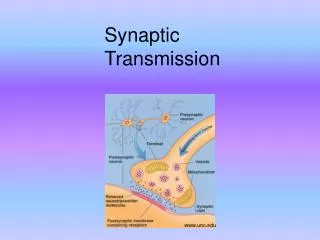
Synapse structure • Presynaptic – transmitting information • Postsynaptic – receiving information • Synaptic terminal protrudes from axon of presynaptic cell • Usually small size (100 – 500 nm across) • Contains vesicles of neurotransmitter ligand • Postsynaptic density on postsynaptic cell dendrite contains neurotransmitter receptors • Ionotropic (change ions) receptors are ligand-gated ion channels • Metabotropic (change metabolism) receptors initiate intracellular signaling cascades • Presynaptic terminal and postsynaptic density separated by very small (10 – 20 nm) synaptic cleft • This minimizes transmission time from presynaptic → postsynaptic
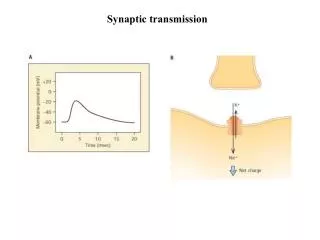
Presynaptic release of neurotransmitter • Neurotransmitter release initiated by AP propagating into presynaptic terminal • Large transient depolarization of presynaptic terminal opens voltage-gated Ca2+ channels • Increase in intracellular free Ca2+ initiates a cascade of events that result in exocytosis of vesicles containing neurotransmitter • Vesicle membranes contain specialized Ca2+ binding proteins • SNARE proteins facilitate docking of vesicles on inner surface of plasma membrane • Ca2+ binding protein synaptotagmin initiates fusion of vesicle and plasma membrane for exocytosis
Presynaptic release of neurotransmitter • Higher presynaptic [Ca2+]i increases rate of exocytosis until saturation • [Ca2+]i increases with number and rate of APs traveling toward presynaptic terminal • Neurotransmitter ligand diffuses across synaptic cleft and binds to postsynaptic receptors • Ligand binding is terminated by enzymatic breakdown within cleft or active reuptake of neurotransmitter molecules by neighboring cells • Many psychiatric and psychotropic drugs function to prevent neurotransmitter uptake or breakdown • Prolongs ligand - receptor binding by maintaining ligand concentration in synaptic cleft
How does a depolarization from a presynaptic AP lead to the release of neurotransmitter? • Ca2+ influx through voltage-gated Ca2+ channels • K+ influx through voltage-gated K+ channels • Na+ influx through voltage-gated Na+ channels • Vesicle docking proteins are voltage-gated
Postsynaptic potentials • Neurotransmitter binding can open ligand-gated ion channel on postsynaptic density • Resulting ionic flux can be depolarizing or hyperpolarizing depending on ionic species that permeates open channels • EPSP – excitatory postsynaptic potential • Depolarization due to opening of Na+ or Ca2+ permeant ligand-gated ion channels • Termed “excitatory” since Vm of postsynaptic cell is pushed closer to AP threshold
Postsynaptic potentials • IPSP – inhibitory postsynaptic potential • Hyperpolarization due to opening of K+ or Cl- permeant ligand-gated ion channels • Termed “inhibitory” since Vm of postsynaptic cell is pushed farther from AP threshold • PSPs – general term for both EPSPs and IPSPs • Characteristic time course due to diffusion, binding and unbinding of neurotransmitter ligand • Desensitization - closing of ligand-gated ion channel while ligand is still bound to receptor • Similar to inactivation of voltage-gated Na+ channel during depolarization • Requires removal of ligand before channel can open again
A neurotransmitter activates a ligand-gated K+ channel. This should produce: • An EPSP • An IPSP • Both • Neither
Synaptic integration • In most neurons, a single EPSP will not drive postsynaptic Vm past AP threshold • Postsynaptic APs are typically evoked by simultaneous synaptic inputs from convergent sources • Temporal summation – rapid EPSPs from same presynaptic terminal • Example: repetitive activation of terminal labeled “A” • Spatial summation – simultaneous EPSPs from different presynaptic terminals • Example: simultaneous activation of terminals labeled “A” and “B” • IPSPs will serve to negate EPSPs or drive Vm below AP threshold
Synaptic strength • Unlike APs, PSPs are graded and can vary in amplitude and time course • Presynaptic factors affecting PSP amplitude and time course • Rate of neurotransmitter synthesis • Amount of neurotransmitter per vesicle • Amount of Ca2+ entry per presynaptic AP • Number of vesicles • Up or down regulation of neurotransmitter release via intracellular signaling molecules • Synaptic cleft factors affecting PSP amplitude and time course • Cleft geometry and neurotransmitter diffusion • Uptake or breakdown of neurotransmitters • Postsynaptic factors affecting PSP amplitude and time course • Spatial or temporal summation of PSPs • Number of neurotransmitter receptors • Up or down regulation of neurotransmitter receptors via intracellular messengers
Synaptic strength • Many drugs called neuromodulators act to modulate neurotransmitter release • Other neuromodulators prevent activation of neurotransmitter receptor by ligand • Many presynaptic terminals have axo-axonic synapses to modulate neurotransmitter release • Many presynaptic terminals have autoreceptors that bind to transmitters released from same terminal • Serves as negative feedback to prevent excess release of neurotransmitter • Many diseases affect synaptic transmission • Tetanus – bacterial toxin that destroys proteins involved in inhibitory neurotransmitter release • Most toxins from venomous species are potent antagonists of voltage- and ligand-gated ion channels • Paralyze or kill prey by preventing APs or synaptic transmission
A drug increases an EPSP produced by a pre-synaptic input. This drug could be acting by: • Increased reuptake of the neurotransmitter • Increased Ca2+ influx through voltage-gated Ca2+ channel • Presynaptic autoreceptor that decreases vesicle docking • Postsynaptic GPCR that decreases response of ligand-gated ion channel
Neurotransmitters • -ergic refers to the type of neurotransmitter a neuron releases • Acetylcholine (ACh) and cholinergic neurotransmission • Primary excitatory neurotransmitter in PNS • Used by somatic and preganglionic autonomic neurons • Degraded by enzyme acetylcholinesterase • Nerve gas Sarin inhibits acetylcholinesterase • Catecholamines – derivatives of tyrosine • Includes dopamine and epinephrine • Broken down by monoamine oxidase (MAO) • MAO inhibitors used to treat psychiatric disorders • Dopamine linked to Parkinson’s disease • Epinephrine (adrenaline) and norepinephrine (noradrenaline) regulate heart rate and blood pressure
Neurotransmitters • Other biogenic amines • Called biogenic amines due to synthesis from amino acid precursors • Serotonin or 5-hydroxytryptophan (5-HT) associated with alertness, appetite, emotional state • Prozac blocks 5-HT uptake, LSD blocks 5-HT receptors • Histamine associated with immune and injury responses • Antihistamines to prevent cold symptoms and inflammation • Amino acids • Major source of excitatory and inhibitory neurotransmitters in brain • Glutamate receptors (GluRs) are majority of excitatory ligand-gated ion channels in brain • Glycine and GABA (g-amino butyric acid) are majority of inhibitory ligand-gated ion channels in brain • Many drugs including barbiturates and benzodiazepines (Valium) act at GABA receptors
Neurotransmitters • Neuropeptides – small polypeptides • Mainly involved in pain sensation and analgesia • Opiate receptors target of morphine and codeine • Other neurotransmitters • ATP can act as neuromodulator • Diffusable gases nitric oxide and carbon monoxide act at intracellular receptors • Not classical neurotransmitter because they don’t require vesicles to be released • Release is of nitric oxide and carbon monoxide is driven by presynaptic production