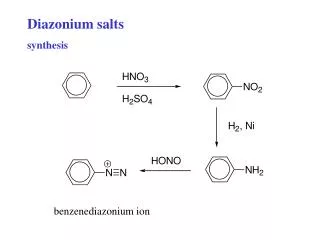
Diazonium salts synthesis
Diazonium salts synthesis. benzenediazonium ion. Diazonium salts, reactions Coupling to form azo dyes Replacements a) -Br, -Cl, -CN b) -I c) -F d) -OH e) -H f) etc. coupling to form azo dyes. Sandmeyer. Ar-N 2 + + CuCl Ar-Cl + N 2


Diazonium salts synthesis
E N D
Presentation Transcript
Diazonium salts synthesis benzenediazonium ion
Diazonium salts, reactions • Coupling to form azo dyes • Replacements • a) -Br, -Cl, -CN • b) -I • c) -F • d) -OH • e) -H • f) etc.
Sandmeyer Ar-N2+ + CuCl Ar-Cl + N2 Ar-N2+ + CuBr Ar-Br + N2 Ar-N2+ + CuCN Ar-CN + N2
Ar-N2+ + KI Ar-I + N2 Ar-N2+ + HBF4 Ar-F + N2 Ar-N2+ + H2O, H+ Ar-OH + N2 Ar-N2+ + H3PO2 Ar-H + N2
In syntheses, you may no longer assume that you can separate a pure para isomer from an ortho/para mixture. Either look up the physical properties of the compounds or rely on experience gained in the homework as to which mixtures are separable and which ones are not!
Spectroscopy of amines Infrared: N—H stretch 3200 – 3500 cm-1 1o often two bands 2o one band 3o no bands N—H bend 1o strong bands 650-900 cm-1(broad) and 1560-1650 cm-1 nmr: N—H 1-5 ppm (often broad and low)
p-toluidine N—H stretch N—H bend
p-ethylaniline d c b a