Salts
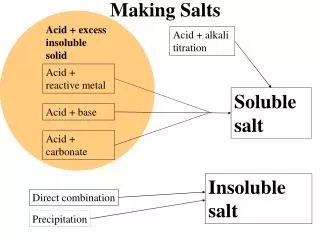
Salts. Since acid + base salt + H 2 O, when a salt is mixed in water, it dissociates. The cation and anion may be able to absorb H + and OH - ions. This could alter the pH of a solution. To determine if a salt solution is acidic or basic Divide the salt into cation and anion

Salts
E N D
Presentation Transcript
Salts • Since acid + base salt + H2O, when a salt is mixed in water, it dissociates. The cation and anion may be able to absorb H+ and OH- ions. This could alter the pH of a solution. • To determine if a salt solution is acidic or basic • Divide the salt into cation and anion • 2. Recreate the acid and base using H+ and OH- • 3. Compare the strengths of the acid and base strong strong neutral 7 strong weak acidic low weak strong basic high weak weak ????? ????
For weak acids and bases, we look at Ka and Kb of each. The higher value determines if the solution is acidic or basic. Reminder Strong acids HCl HBr HI HNO3 HClO3 HClO4 H2SO4 Strong Bases Group I hydroxides Heavy group 2 hydroxides Example – Determine if the following solutions should be acidic or basic. a. CaCl2 b. NaNO3 c. Na2SO3 d. KC2H3O2 e. NH4Br neutral basic basic basic acidic
Determining the pH of a salt solution • Split the salt into its acid and base ions • 2. Ignore the strong one (this will not alter the [H+] or [OH-]) • 3. Treat the cation as a weak acid and the anion as a weak base and write the Ka or Kb expression • 4. Use Ka or Kb to determine the concentration of H+ or OH- • 5. Determine pH Examples – Write out the important equilibrium that occurs when we add the following salts to water a. NaClO b. NH4Cl c. KNO2 • ClO- + H2O • NH4+ + H2O • NO2- + H2O HClO + OH- H3O+ + NH3 HNO2 + OH-
Examples If Ka HF = 6.8 x 10-4 and Ka of NH4+ is 5.6 x 10-11, determine the pH of a. 0.15 M NH4Cl solution b. 0.50 M NaF solution a. Since NH4Cl will be acidic, treat NH4+ as a weak acid, ignore Cl-. NH4+ NH3 + H+ 0.0 M 0.15 M 0.0 M -x +x +x 0.15 x x Ka = [NH3][H+] [NH4+] 5.6 x 10-11 = x2 0.15 X = 2.90x 10-6 pH = -log 2.90x 10-6 pH =5.54
For problem B, NaF Na+ + F- ignoretreat as a weak base F- + H2O HF + OH- 0.0 M 0.5 M 0.0 M -x +x +x 0.5 x x We need Kb for F-, so alter the Ka Ka x Kb = Kw 6.8 x 10-4 x Kb = 1 x 10-14 Kb = 1.47 x 10-11 Kb = [HF][OH-] [F-] 1.47 x 10-11 = x2 0.5 X = 2.71x 10-6 pOH = -log 2.71x 10-6 pOH =5.56 pH = 8.44