Integrated rate laws
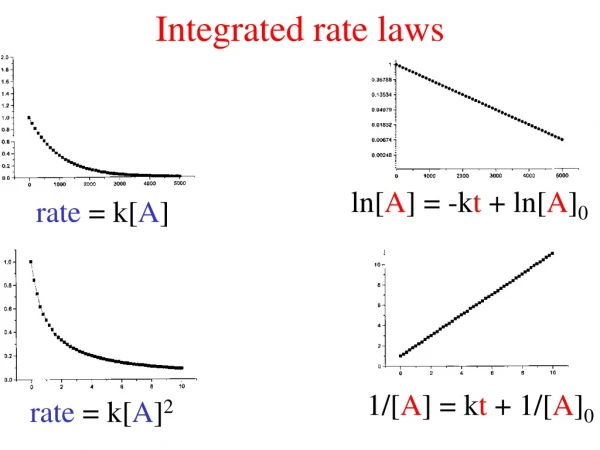
Integrated rate laws. ln[ A ] = -k t + ln[ A ] 0. rate = k[ A ]. 1/[ A ] = k t + 1/[ A ] 0. rate = k[ A ] 2. Reaction mechanism. Elementary steps. reaction =. Molecularity. Elementary step. rate law. uni molecular . A product . k[ A ]. A + B product . bi molecular .

Integrated rate laws
E N D
Presentation Transcript
Integrated rate laws ln[A] = -kt + ln[A]0 rate = k[A] 1/[A] = kt + 1/[A]0 rate = k[A]2
Reaction mechanism Elementary steps reaction = Molecularity Elementary step rate law unimolecular A product k[A] A+ B product bimolecular k[A][B] A+B+C product k[A][B][C] termolecular A+A+A product k[A]3 chemical reaction = sum of elementary steps rate law and stoichiometry
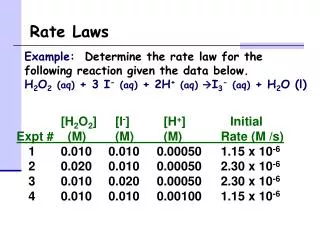
Reaction mechanism 2H2O2 (aq) 2H2O(l) + O2(g) Gorxn = [ 2(-237.9)] - [2(-131.67)] = -212.46 kJ spontaneous reaction experimentalrate law: rate = k[H2O2] [I-] I- = catalyst increase rate of reaction not consumed in the overall reaction reactant in early elementary step product in later elementary step
Reaction mechanism 2H2O2(aq) 2H2O(l) + O2(g) rate = k[H2O2] [I-] k1 step 1 H2O2 H2O + OI- + I- k2 step 2 H2O2 + OI- H2O + O2 +I- 2H2O2(aq) 2H2O(l) + O2(g) step 1 rate = k1 [H2O2] [I-]
Reaction mechanism k1 H2O2 + I- H2O + OI- step 1 k2 H2O2+ OI- H2O + O2 +I- step 2 what about step 2 ? assume k2 >> k1 step 1 rate determining step I- catalyst consumed in rate determining step regenerated in later elementary step intermediate OI- formed in early step, consumed in later step
Rate determining step on Labor Day weekend “Big Mac” bridge
Reaction Mechanism H2(g) + I2(g) 2HI(g) rate = k [H2] [I2] reaction faster in light free radical unpaired electron
Reaction Mechanism H2(g) + I2(g) 2HI(g) rate = k [H2] [I2] I2 2I. step 1 forward rate = kf [I2] kr [I.]2 reverse rate = kf[I2] = kr[I.]2 equilibrium = kf Keq= [I.]2 [I2] kr
I22I. Reaction Mechanism H2(g) + I2(g) 2HI(g) rate = k [H2] [I2] step 1 step 2 H2+ 2I. 2HI 2HI H2 + I2 need H2in the rate determining step from step 2 rate = k [H2] [I.]2 I.= intermediate
I22I. Reaction Mechanism H2(g) + I2(g) 2HI(g) rate = k [H2] [I2] step 1 step 2H2+ 2I. 2HI = kf rate = k [H2] [I.]2 [I.]2 Keq = [I2] kr Keq[I2] rate = k [H2] [I.]2 = Keq [I2] rate = k’ [H2] [I2]
2NO N2O2 Reaction Mechanism 2NO + O2 2NO2 rate = k[NO]2 step 1 N2O2 + O2 2NO2 step 2 2NO + O2 2NO2 overall reaction: intermediates: N2O2 relative rates of steps 1 and2
2NO N2O2 2NO + O2 2NO2 rate = k[NO]2 step 1 N2O2 + O2 2NO2 step 2