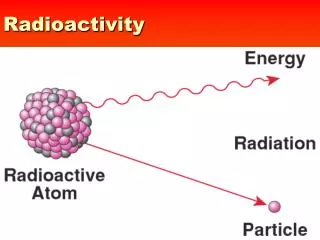
Radioactivity
Radioactivity. 1) Introduction 2) Decay law 3) Alpha decay 4) Beta decay 5) Gamma decay 6) Fission 7) Decay series 8) Exotic forms of decay. Detection system GAMASPHERE for study rays from gamma decay. Introduction.

Radioactivity
E N D
Presentation Transcript
Radioactivity 1) Introduction 2) Decay law 3) Alpha decay 4) Beta decay 5) Gamma decay 6) Fission 7) Decay series 8) Exotic forms of decay Detection system GAMASPHERE for study rays from gamma decay
Introduction Transmutation of nuclei accompanied by radiation emissions was observed - radioactivity. Discovery of radioactivity was made by H. Becquerel (1896). Three basic types of radioactivity and nuclear decay: 1) Alpha decay 2) Beta decay 3) Gamma decay and nuclear fission (spontaneous or induced) and further, more exotic types of decay Transmutation of one nucleus to another proceed during decay (nucleus is not changed during gamma decay – only energy of excited nucleus is decreased). Mother nucleus – decaying nucleusDaughter nucleus – nucleus incurred by decay Sequence of followup decays – decay series. Decay of nucleus is not dependent on chemical and physical properties of nucleus neighboring (exception is for example gamma decay through conversion electrons which is influenced by chemical binding). Electrostatic apparatus of P. Curie for radioactivity measurement (left) and present complex for measurement of conversion electrons (right)
ln N – ln N0 = -λt Radioactivity decay law Activity (radioactivity) A: where N is number of nuclei at given time in sample [Bq = s-1,Ci =3.7·1010Bq]. Constant probability λ of decay of each nucleus per time unit is assumed. Number dN of nuclei decayed per time dt: dN = -Nλdt Both sides are integrated: Then for radioactivity we obtain: where A0 ≡ -λN0 Probability of decay λ is named decay constant. Time of decreasing from N to N/2 is decay half-life T1/2. We introduce N = N0/2: Mean lifetimeτ: For t = τ activity decreases to 1/e = 0,36788. Heisenberg uncertainty principle: ΔE·Δt ≈ ħ → Γ · τ ≈ ħ where Γ is decay width of unstable state: Γ = ħ /τ = ħ λ
Total probability λ for more different alternative possibilities with decay constants λ1,λ2,λ3 … λM: Sequence of decay we have for decay seriesλ1N1 → λ2N2 → λ3N3 → … → λiNi → … → λMNM Time change of Ni for isotope i in series: dNi/dt = λi-1Ni-1 - λiNi We solve system of differential equations and assume: … For coefficients Cij it is valid: i ≠ j Coefficients with i = j can be obtained from boundary conditions in time t = 0: Ni(0) = Ci1 + Ci2 + Ci3 + … + Cii Special case forτ1 >> τ2,τ3 … τM : each following member has the same number of decays per time unit as first. Number of existed nuclei is inversely dependent on its λ. → decay series is inradioactive equilibrium. Creation of radioactive nuclei with constant velocity – irradiation using reactor or accelerator. Velocity of radioactive nuclei creation is P: dN/dt = - λN + P Solution of equation(N0 = 0): λN(t) = A(t) = P(1 – e-λt) It is efficient to irradiate number of half-lives but not so long time – saturation starts. Development of activity during homogenous irradiation
Alpha decay High value of alpha particle binding energy → EKIN sufficient for escape from nucleus → Relation between decay energy and kinetic energy of alpha particles: Decay energy: Q = (mi – mf –mα)c2 Kinetic energies of nuclei after decay (nonrelativistic approximation): EKIN f = (1/2)mfvf2 EKIN α = (1/2) mαvα2 From momentum conservation law: mfvf = mαvα → ( mf >> mα → vf << vα) From energy conservation law: EKIN f + EKIN α = Q (1/2) mαvα2 + (1/2)mfvf2 = Q We modify equation and we introduce: Kinetic energy of alpha particle: Typical value of kinetic energy is 5 MeV. For example for 222Rn: Q = 5.587 MeV andEKIN α= 5.486 MeV. Barrier penetration: Particle (Z,A) impacts on nucleus (Z,A) – necessity of potential barrier overcoming. For Coulomb barier is the highest point in the place, where nuclear forces start to act:
bound state quasistationary state Barrier height is VCB ≈ 25 MeV for nuclei with A=200. Problem of penetration of α particle from nucleus through potential barrier → it is possible only because of quantum physics. Assumptions of theory of α particle penetration: 1) Alpha particle can exist at nuclei separately 2) Particle is constantly moving and it is bonded at nucleus by potential barrier 3) It exists some (relatively very small) probability of barrier penetration. Probability of decay λ per time unit: λ = νP where ν is number of impacts on barrier per time unit and P probability of barrier penetration. We assumed, that α particle is oscillating along diameter of nucleus: Probability P = f(EKINα/VCB). Quantum physics is needed for its derivation. Centrifugal barrier depends on angular momentum of emitted or incident particle: classical: quantum: L2 → l(l+1)ħ2 →
Relative electron intensity Electron energy Beta decay Nuclei emits electrons: 1) Continuous distribution of electron energy (discrete was assumed – discrete values of energy (masses) difference between mother and daughter nuclei). Maximal EEKIN = (Mi – Mf – me)c2. 2) Angular momentum – spins of mother and daughter nuclei differ mostly by 0 or by 1. Spin of electron is but 1/2 → half-integral change → postulation of new particleexistence – neutrino. mn > mp + mν→ spontaneous process neutron decay τ ≈ 900 s (strong ≈ 10-23 s, elmg ≈ 10-16 s)→ decay is made by weak interaction inverse process proceeds spontaneously only inside nucleus Process of beta decay – creation of electron (positron) or capture of electron from atomic shell accompanied by antineutrino (neutrino) creation inside nucleus. Z is changed by one. A is not changed. Schematic dependence Ne = f(Ee) at beta decay
According to mass of atom with chargeZwe obtain three cases: 1) Mass is larger than mass of atom with charge Z+1 → electron decay – decay energy is split between electron a antineutrino, neutron is transformed to proton: 2) Mass is smaller than mass of atom with charge Z+1 but it is larger than mZ+1 – 2mec2 → electron capture – energy is split between neutrino energy and electron binding energy. Proton is transformed to neutron: 3) Mass is smaller than mZ+1 – 2mec2 → positron decay – part of decay energy higher than 2mec2 is split between kinetic energies of neutrino and positron. Proton changes to neutron: Discrete lines on continuous spectrum : 1) Auger electrons – vacancy after electron capture is filled by electron from outer electron shell and obtained energy is realized through Röntgen photon. Its energy is only a few keV → it is very easy absorbed → complicated detection 2) Conversion electrons – direct transfer of energy of excited nucleus to electron from atom Beta decay can goes on different levels of daughter nucleus, not only on ground but also on excited. Excited daughter nucleus then realized energy by gamma decay. Some mother nuclei can decay by two different ways either by electron decay or electron capture to two different nuclei. During studies of beta decay discovery of parity conservation violation in the processes connected with weak interaction was done.
Relative electron intensity Electron energy Neutrino – particle interacted only by weak interaction, very small cross-section. Detection by inverse beta decay: Determination of neutrino mass from form of end of electron spectra We can express function connected to dependency of number of electrons on their energy: where N(Ee) – number of electrons, F*(Z,Ee) – Fermi function containing of correction on Coulomb field of nucleus and electron cloud. In the case on nonzero neutrino mass : EMAX=Q - mνc2. (Q – decay energy).The graph of dependency is named as: Fermi graph – possibility of accurate determination of maximal energy (decay energy) – eventually neutrino mass. The neutrino mass determined by tritium decay is 2 eV at present time. Tests and transport of main vacuum chamber of KATRIN spectrometer Fermi graph for decay of tritium 3H
Gamma decay Excited nucleus unloads energy by photon irradiation After alpha or beta decay → daughter nuclei at excited state → emission of gamma quantum → gamma decay Multipole expansion and simple selective rules: Different transition multipolarities: Electric EJ → spin I = min J, parity π = (-1)I Magnetic MJ → spin I = min J, parity π = (-1)I+1 Transition between levels with spinsIiandIfand paritiesπiandπf: I = |Ii – If| pro Ii≠ If I = 1 for Ii = If > 0 π = (-1)I+K = πi·πf K=0 for E and K=1 pro M Electromagnetic transition with photon emission between states Ii = 0 andIf = 0 does not exist Energy of emitted gamma quantum: Eγ = hν = Ei - Ef More accurate (inclusion of recoil energy): Momenta conservation law → hν/c = Mjv Energy conservation law → where ΔER is recoil energy.
Level width Γ is connected with its live time by Heisenberg uncertainty principle: Γτ ħ And then Γ ħ/τ ~ uncertainty of (Ei – Ef). Nucleus can be excited by the same energy Eγ which can emit. Nucleus recoil must be included (recoil is created also during absorption): Γ ≥ 2∙ ΔER for possibility of resonance absorption. This is right for free atom. Transition Eγ = 14 keV for isotope 57Fe: For level τ ~ 10-7 s → Γ ~ 10-8 eV and ΔER ~ 10-3 eV. → Γ << ΔER Atom bounded in crystal lattice → momentum is transferred to whole crystal lattice → little energy transfer → possibility of resonance absorption – Mössbauer phenomena. Mössbauer phenomena makes possible very accurate measurements of level energy and width. We have: 1) Source of gamma quanta 2) Moving absorber 3) Detector of gamma rays Doppler phenomena changes with absorber velocity energy of gamma quantum by ΔE = E∙v/c, we can measure intensity of absorption → form of Mössbauer lines is visible.
Mean lifetimes of levels are mostly very short ( < 10-7s – electromagnetic interaction is much stronger than weak) → life time of previous beta or alpha decays are longer → time behavior of gamma decay reproduces timebehavior of previous decay. They exist longer and even very long life times of excited levels - isomer states. Probability (intensity) of transition between energy levels depends on spins and parities of initial and end states. Usually transitions, for which change of spin is smaller, are more intensive. System of excited states, transitions between them and their characteristics are shown by decay schema. Example of part of gamma ray spectra from source 169Yb → 169Tm: Decay schema of 169Yb → 169Tm:
Internal conversion Direct transfer of energy of excited nucleus to electron from atomic electron shell (Coulomb interaction between nucleus and electrons): Energy of emitted electron: Ee = Eγ – Be where Eγ is excitation energy of nucleus, Be is binding energy of electron Alternative process to gamma emission. Total transition probability λ is: λ = λγ + λe The conversion coefficientsα are introduced: It is valid: dNe/dt = λeN and dNγ/dt = λγN and then: Ne/Nγ = λe/λγ and λ = λγ (1 + α) where α = Ne/Nγ We label αK, αL, αM, αN, … conversion coefficients of corresponding electron shell K, L, M, N, …: α = αK + αL + αM + αN + … The conversion coefficients decrease with Eγ and increase with Z of nucleus. Transitions Ii = 0 → If = 0: only internal conversion not gamma transition The place freed by electron emitted during internal conversion is filled by other electron and Röntgen ray is emitted with energy: Eγ = Bef - Bei characteristic Röntgen rays of correspondent shell. Energy released by filling of free place by electron can be again transferred directly to another electron and the Auger electronis emitted instead of Röntgen photon. Pair internal conversion – Eγ > 2mec2 → pair electron and positron pair can be created → it is not connected to electron shell → probability increases with Eγ.
Nuclear fission The dependency of binding energy on nucleon number shows possibility of heavy nuclei fission to two nuclei (fragments) with masses in the range of half mass of mother nucleus. Penetration of Coulomb barrier is for fragments more difficult than for alpha particles (Z1, Z2 > Zα = 2) →the lightest nucleus withspontaneous fissionis 232Th. Example of fission of 236U: Energy released by fission Ef ≥ VC → spontaneous fission We assume A1=A2=A/2 a Z1=Z2=Z/2. Then magnitude of Coulomb potential barrier is: For fission energy is valid: Ef/c2 = m(Z,A) – 2m(Z/2,A/2) After substitution from Weizsäker formula: Ef/ c2 = aSA2/3(1-21/3)+aCZ2A-1/3(1-2-2/3) → Ef = (1-21/3) c2aSA2/3+(1-2-2/3) c2aCZ2A-1/3 = aS‘A2/3+aC‘Z2A-1/3 = A2/3 (aS‘+aC‘Z2/A) From this: Ef > 0 → Z2/A > -aS‘/aC‘~ 18 Ef ≥ VC Ratio Z2/A (fission parameter) is critical for stability against spontaneousfission.
After supplial of energy – induced fission – energy supplied by photon (photofission), by neutron, … Induced fission can be described by comparison of surface and Coulomb energy of symmetrical sphere and deformed ellipsoid with half-axe a and b with the same volume V: The same volume → the same volume energy for sphere and ellipsoid.We express a = R(1- ε) and b = R(1-ε)-1/2 where ε is ellipsoid eccentricity. Ellipsoid surface is: Surface energy in Weizsäker formula then is: Coulomb energy of charged ellipsoid: Then from Weizsäker formula: Deformation energy ED (ΔES and ΔEC differences between energies of ellipsoid and sphere ε = 0): After substitution of constants from Weizsäker formula: ED = ε2(7.34∙A2/3 –0.14∙Z2A-1/3) = ε2∙A2/3 (7.34–0.14∙ Z2/A) [MeV] Z2/A ≥ 51 → ED ≤ 0 →spontaneous fission
Energy Ea needed for overcoming of potential barrier – activation energy – for heavy nuclei is small ( ~ MeV) → energy released by neutron capture is enough (high for nuclei with odd N). Certain number of neutrons is released after neutron capture during each fission (nuclei with average A have relatively smaller neutron excess than nucley with large A) → further fissions are induced → chain reaction. 235U + n → 236U → fission → Y1 + Y2 + ν∙n Average number η of neutrons emitted during one act of fission is important - 236U (ν = 2.47) or per one neutron capture for 235U (η = 2.08) (only 85% of 236U makes fission, 15% makes gamma decay). How many of created neutrons produced further fission depends on arrangement of setup with fission material Ratio between neutron numbers in n and n+1 generations of fission is named as multiplication factork: Its magnitude is split to three cases: k < 1 – subcritical– without external neutron source reactions stop →accelerator driven transmutors – external neutron source k = 1 – critical– can take place controlled chain reaction →nuclear reactors k > 1 – supercritical– uncontrolled (runaway) chain reaction →nuclear bombs Fission products of uranium 235U.Dependency of their production on mass number A:(taken from A. Beiser: Concepts of modern physics)
Decay series Different radioactive isotope were produced during elements synthesis (before 5 – 7 miliards years). Some survive: 40K, 87Rb, 144Nd, 147Hf The heaviest from them: 232Th, 235U a 238U Beta decay: Ais not changedAlpha decay: A → A - 4 Summary of decay series: Decay life time of mother nucleus of neptunium series is shorter than time of Earth existence. Also all furthers → must be produced artificially → with lower A by neutron bombarding, with higher A by heavy ion bombarding. Some isotopes in decay series must decay by alpha as well as beta decays → branching Possibilities of radioactive element usage: 1) Dating (archeology, geology) 2) Medicine application (diagnostic – radioisotope, cancer irradiation) 3) Measurement of tracer element contents (activation analysis) 4) Defectology, Röntgen devices
Exotic forms of decay Proton emission – protons must penetrate Coulomb barrier → life time (also in μs and ms range) is longer than characteristic nuclear time (time of nucleon transit through nucleus – 10-21s) → existence of proton radioactivity. It is possible only for exotic light nuclei with large excess of protons (for example 9B) – decay has sufficiently short decay time and hence it is not suppressed by competitive positron beta decay. Emission of couple of protons – made by coupling (maybe also in form of 2He) - year 2000 at Oak Ridge laboratory for nucleus 18Ne Delayed proton emission – emission of protons following after proton decay → nuclei with large excess of protons → created nucleus at highly excited state emits proton Neutron emission– life time of nuclei with big neutron excess, if neutron decay is energy possible, is comparable with characteristic nuclear time – it can not be named as neutron radioactivity Delayed neutron emission following after beta decay. Nucleus with big neutron excess → beta decay with longer decay time → consecutive quick neutron emission during time comparable with characteristic nuclear time. Emission of hevier nuclei – 12C, 16O … → fragmentation of highly excited nuclei
Double beta decay (ββ2ν) – is possible if double decay is allowed by energy conservation and single beta decay is not allowed. We have potentially 35 (ββ 2ν) – emitters. Nine was observed up to now (48Ca, 76Ge, 82Se, 100Mo, …). Very long decay time T1/2 = 1019 – 1024 years. Studied using underground experiments (main problem is background). For example new device NEMO-3 (10 kg of 100Mo, Qββ = 3.038 MeV). Next possibility – geochemical measurements. Examples of nuclei decayed by double beta decay
Neutrinoless double beta decay (ββ0ν) – possible only in the case of nonzero neutrino rest mass and if neutrino is Majorana particle type (antiparticle is identical with particle – difference of lepton number is still there). In this case two neutrons can change neutrino and antineutrino in the process violated lepton number conservation and only pair of electrons is emitted. This decay was not observed up to now .Limit is in the range of 1025 years measured with 76Ge → limit on mass ~ 0.45 eV. Device NEMO-3for double beta studies Device NEMO-3 in underground tunnel at Alps