Radioactivity
Radioactivity. Prepared by: Timothy John D. Matoy. RADIOACTIVITY. Process of loosing energy to reach a stable state. Radioactivity and Radioactive Decay. The nuclei of some nuclides are not stable

Radioactivity
E N D
Presentation Transcript
Radioactivity Prepared by: Timothy John D. Matoy
RADIOACTIVITY • Process of loosing energy to reach a stable state.
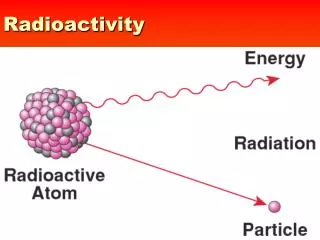
Radioactivity and Radioactive Decay • The nuclei of some nuclides are not stable • They disintegrate or undergo nuclear transformation spontaneously and in random process called radioactivity
Henri Becquerel (1852-1908) • Discover the radioactivity in 1896 • He noted that a piece of mineral containing uranium when placed over an exposed photographic plate just as if it has been exposed to light.
Marie Curie (1867-1924) • Pierre and Marie Curie discovered that polonium and radium also emit radiation.
Artificial Radioactivity • Radioactivity produced by man • Irene Curie-Joliot produce the first radioactive product when they bombarded aluminum with alpha particles from polonium source to study the emitted neutrons and positrons.
Radioactive elements emitted into 3 types of radiation • Alpha Particles • Beta Particles • Gamma Rays
Alpha Particles • Fast moving helium nuclei; positive electrical charge
Beta Particles • Negative electrical charged electrons.
Electrons, Positrons • Very light; in tissue do not travel in straight lines but are deflected by coulombic repulsions from atomic orbital electrons • Lose an average 50% of their energy in interaction
Gamma Rays • Electromagnetic waves of very short wavelength and travelling within the speed of light. • No charge at all.
Protons • Generated by cyclotron beams • Because they are heavier than electrons, travel mainly in straight line by boring a path through atomic clouds • Medium Z materials used for shielding
Half-Life(t½) • the time in which a radioactive substance will lose half of its activity through disintegration. • Physical Half-life • Biological Half-life • Effective Half-Life
Physical Half-Life • the average time required for the decay of half the atoms in a given amount of a radioactive substance.
Biologic Half-Life • the time in which a living tissue, organ, or individual eliminates, through biologic processes, half of a given amount of a substance that has been introduced into it.
Effective Half-Life • the half-life of a radioactive isotope in a biologic organism, resulting from the combination of radioactive decay and biologic elimination.
Types of Decay • Alpha Decay • Beta Negative Decay • Beta Positive Decay • Gamma Ray Emission • Electron Capture
BETA DECAY • Beta minus • Electron antineutrino • Interacts with neutron • Beta plus • Electron Neutrino • Interacts with protons
ELECTRON CAPTURE • Electron capture is a process in which a proton-rich nuclide absorbs an inner atomic electron (changing a nuclear proton to a neutron) and simultaneously emits a neutrino.
Radioactive Decay Law • Elster and Geitel observed that the strength of a pure radioactive substance decrease exponentially. • Radioactivity was found to be a property of the individual atoms, not of a substance as a whole.
Unit of Radioactivity • Curie (Ci) • Becquerel (Bq) • 1 Bq = 1 disintegration per second • 1 Ci = 3.7x1010Bq
Exponential decay law expressed in the following equation: • N = N0e-λt or A = A0e-λt Where: • A = present activity • A0 = original activity • λ = disintegration constant/ decay constant • t = elapsed time
Sample Problem • The half life for radioactive radon gas is 3.83 days, what will be the present activity of the radon gas after 5 days if the initial activity is 30 mCi?
Practice Problem • A Cesium-138 radioactive source has a half life of 30 years. If the initial activity is 10.25 Ci. What will be the present activity after 30 years?
Practice Problem • A certain radioactive source has a present activity of 2.1626 mCi after 47.5 days. If the initial activity is 15 mCi. Find the decay constant?
Activity Fraction • the fraction of the activity that is remaining after a given amount of time. • AF = 2 –n • Where n = time/ half life
Sample Problem • The half life for radioactive radon gas is 3.83 days, what will be the its activity fraction after 5 day?
Practice Problem • A Cesium-138 radioactive source has a half life of 30 years. If the initial activity is 10.25 Ci. What will be its activity fraction after 55 years?
With the activity fraction, we can write a single equation to solve for the final activity if we are given the initial activity • Nf= (AF) Ni • Where Nf= final activity, Ni = initial activity
Sample Problem • The half life for radioactive radon gas is 3.83 days, what will be the present activity of the radon gas after 5 days if the initial activity is 30 mCi?
Practice Problem • A Cesium-138 radioactive source has a half life of 30 years. If the initial activity is 10.25 Ci. What will be the present activity?
Radioactive Decay Law Activity Remaining = original activity (0.5)n • Where n – number of half-lives
Sample Problem • The half life for radioactive radon gas is 3.83 days, what will be the present activity of the radon gas after 5 days if the initial activity is 30 mCi?
Practice Problem • A Cesium-138 radioactive source has a half life of 30 years. If the initial activity is 10.25 Ci. What will be the activity after 30 years?
PRACTICE PROBLEM • A certain radioactive source has an initial activity of 10.25 Ci. After 30 years only 5.125 Ci of activity remains. What is its half-life?
Practice problem • Technetium 99m with a half-life of 6 hours was left in the laboratory at 6 o’clock in the morning. If the original activity is 100 mCi, when do the radioactive substance will have a 6.25% of its original value? What will be its activity at that time?