Bonding
Bonding. Chemical bonding. Chemical Bonding. Graphite. Diamond. Silicon dioxide. Ionic Bond / metallic bond / covalent bond. Nature of the bond Physical properties Melting point / boiling point Electrical conductivity Solubility Hardness . Ionic bonding.

Bonding
E N D
Presentation Transcript
Chemical Bonding Graphite Diamond Silicon dioxide
Ionic Bond / metallic bond / covalent bond • Nature of the bond • Physical properties • Melting point / boiling point • Electrical conductivity • Solubility • Hardness
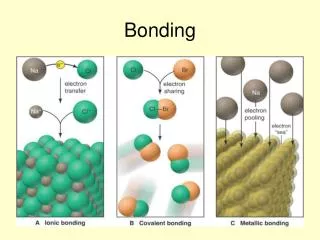
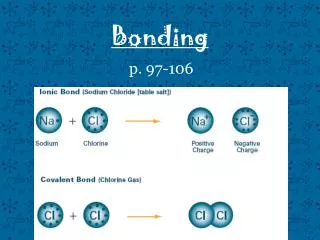
Ionic bonding • electrostatic attraction between oppositely charged ions • The charged ions are formed when atoms lose or gain electrons.
Example Sodium atom Na 2,8,1 Chlorine atom Cl 2,8,7 Sodium ion Na+ 2,8 Chloride ion Cl- 2,8,8 Na x [Na]+ + e- Cl + e- [ Cl ]- Na x + Cl [Na]+[ Cl x]- Sodium metal reacts with chlorine gas in a violent exothermic reaction to produce NaCl.
Formation of ions What ions will be formed by these elements? Na , Mg , Al , P , S and Cl Draw the dot and cross diagram for magnesium oxide
Ions of transition elements • The transition elements form more than one stable positive ion.
Physical properties of ionic compounds • Melting point / boiling point • Electrical conductivity • Solubility • Hardness • Volatility Students’ posters
Hardness • Hard but brittle because a slight displacement of a layer of ions will cause repulsion between the ions and the crystal will break.
Volatility (how readily a substance evaporates) - Low volatility because of the strong electrostatics forces between ions.
PreDicting the type of bonding from electronegativity values • Ionic bonding typically occurs between metal and non-metal. E.g. Barium fluoride, BaF2 • The reactivity of metals and non-metals can be assessed using electronegativity (ability of an atom in a covalent bond to attract shared pairs of electrons to itself).
Metals generally have low electronegativity values, while non-metals have relatively high electronegativity values. • Fluorine, which has the greatest attraction for electrons in bond-forming situations (highest E value).
If the difference in E values is > 1.8 => ionic bond • If the difference in E values is 0, non-polar covalent bond • If the difference is 0 – 1.8, polar covalent bond
Electrons are not shared. E.g. Na+ Cl- , electron is transferred. Polar covalent bonds are covalent bonds with ionic character. Ionic bond Electrons are equally shared. E.g.Cl-Cl Non polar covalent bond Electrons are not equally shared. E.g. Atoms have different electronegativity values Polar covalent bond
Example • Use the table above to predict the type of bonding beween Fluorine , F2 Hydrogen iodide, HI and Lithium fluoride, LiF
Structure of giant ionic compound • In an ionic compound, constituent ions are held in fixed positions in an orderly arrangement by strong ionic bonds. A crystal of NaCl consists of a giant lattice of Na+ and Cl- ions. All Na+ ions in the crystal attract all the Cl- ions, and vice versa. This strong attraction between oppositey charged ions holds the crystal together. It is difficult to break apart the lattice. Page 88
Covalent bonding • forms when atoms share electrons • A covalent bond is the electrostatic attraction between the shared pair of electrons and the nuclei of the atoms making the bond. E.g. Formation of methane molecule, CH4 Bond pair _____ Lone pair . . Lewis Structures of methane Page 90
The bonding pair of electrons spends most of its time between the two atomic nuclei, thereby screening the positive charges from one another and enabling the nuclei to come closer together than if the bonding electrons were absent. Negative charge on the electron pair attracts both nuclei and holds them together in a covalent bond. • When two atoms are chemically bonded, the two atoms close together have less energy and therefore are more stable than when separated. • Energy is given off by the atoms to form a bond, and energy must be supplied (absorbed) to break the bond. • A covalent bond is the result of electrostatic attraction between the nuclei of the 2 atoms and the pair of shared electrons.
Lewis structures Bond pair _____ Lone pair . . Draw Lewis structures of the following molecules: Chlorine, Cl2 Hydrogen chloride, HCl Oxygen, O2 Nitrogen, N2 Carbon dioxide, CO2 Water, H2O Follow structures on page 91
EXceptions • Not all covalent bond results in a noble gas configuration. In BF3, B has only 6 electrons in its outer shell. In SF6, S has 12 electrons in its outer shell (expanded octet) Page 92
Multiple bonds • Bond Strength Triple bonds > Double bonds > Single bonds • The attraction between the 2 nuclei for 3 electron pairs in a triple bond is > that for 2 electron pairs in a double bond which is > than that for 1 electron pair in a single bond. • Triple bonds are shorter due to greater attraction between the bonding electrons and the nuclei with more electrons in the bond. Page 93
Coordinate (dative) bonding • In some molecules and polyatomic ions, both electrons to be shared come from the same atom. The covalent formed is called the coordinate or dative bond. • Carbon monoxide (CO) can be viewed as containing one coordinate bond and two "normal" covalent bonds between the C atom and the O atom. How do you draw the Lewis structure? Page 94
Reaction between ammonia and hydrogen chloride • A thick white smoke of solid ammonium chloride is formed in the reaction below: • Ammonium ions, NH4+, are formed by the transfer of a hydrogen ion from the hydrogen chloride to the lone pair of electrons on the ammonia molecule. Page 94, Lewis acid-base reaction
When the ammonium ion, NH4+, is formed, the fourth hydrogen is attached by a dative covalent bond, because only the hydrogen's nucleus is transferred from the chlorine to the nitrogen. The hydrogen's electron is left behind on the chlorine to form a negative chloride ion. • Once the ammonium ion has been formed it is impossible to tell any difference between the dative covalent and the ordinary covalent bonds.
Dissolving hydrogen chloride gas in water • Something similar happens. A hydrogen ion (H+) is transferred from the chlorine to one of the lone pairs on the oxygen atom. • The H3O+ ion is variously called the hydroxonium ion.
Other examples: • The reaction between ammonia and boron trifluoride, BF3 In BF3, there are only 6 electrons in the outer shell of boron. There is space for the B to accept a pair of electrons.
Draw lewis structures for molecules and ions Rules • Calculate the total no. of valence electrons for all atoms in the molecule or ion. • Divide by 2 to get the no. of electron pairs. • Each electron pair is represented by a line. • Arrange the lines (electron pairs) so that all the atoms are joined together by at least a single bond and the outer atoms have 8 electrons in their outer shell (except for H) • Rearrange the lines (electron pairs) so that every period 2 atom has 4 pairs of electrons. The outer atoms already have 4 pairs, so this should normally only involve moving lone pairs so that they become bonding pairs of eectrons.
Example Write the Lewis structure NF3 , CO32- , NO2- , O3
Bond strength and length of covalent bonds • Strength Triple bonds > Double bonds > Single bonds • Length Single bonds > Double bonds > Triple bonds
Bond polarity • In diatomic molecules (e.g. H2 ,Cl2) both atoms exert an identical attraction. • When the atoms are different (e.g. HCl) with one more electronegative than the other, a polar bond is formed. • Relative polarity is predicted from electronegativity values. • C-O is more polar than C-Cl since the difference in E value for C-O is greater than that for C-Cl.
VSEPR theory • The shapes of simple molecules and ions can be determined by using the Valence Shell Electron Repulsion (VSEPR) theory. • Electron pairs around the central atom repel each other • Bonding pairs and lone pairs arrange themselves to be as far apart as possible
The shape of a molecule depends on the number of electron pairs in the outer shell of the central atom. The 5 basic molecular shapes show the arrangement of the electron pairs (charge centres) that result in minimum repulsion between the bonding and lone pairs of electrons. Focus on 3
Basic shape : Tetrahedral arrangement Tetrahedral arrangement Trigonal pyramidal Bent
How to predict the shapes of molecules? • Draw the Lewis structure for the molecule or ion. • Count up the number of electron pairs (bonding and lone pairs) in the outer shell of the central atom. Multiple bond is counted as single electron pair because the electrons occupy the same space region. • Check the table (page 100) to get the basic shape. Draw the 3-D shapes. • A lone pair is just an electron pair in the outer shell of an atom - cannot be “seen” • State the actual shape of the molecule.
Example Consider CH4 , NH3 , H2O , CO2, SO2
Lone pairs and bond angles Order of repulsion : lone pair-lone pair > lone pair- bonding pair > bonding pair – bonding pair Lone pairs are held closer to the nucleus than the bonding pairs. The distance between the lone pair electrons and the bonding pairs of electrons is shorter than the distance between the bonding pairs to each other. Repulsion due to lone pairs causes the bond angles to become smaller
Order of repulsion : lone pair-lone pair > lone pair- bonding pair > bonding pair – bonding pair Methane, CH4 Bond angle is 109.50 Ammonia, NH3 Greater repulsion by lone pair of electrons. Bond angle is smaller than 109.50(1050) Water, H2O Even greater repulsion by two lone pair of electrons. Bond angle is even smaller (1050)
Predicting the shape of ions Consider NH4+ , H3O+ , NO2- NH4+ As the 4 negative charge centres repel each other and take up positions in space to be as far apart as possible, the electron pairs are distributed in tetrahedral arrangement. H3O+ As the 4 negative charge centres repel each other and take up positions in space to be as far apart as possible, the electron pairs are distributed in tetrahedral arrangement. With one lone pair of electrons, the actual structure is trigonal pyramid with a bond angle of 1070 for H-O-H bond. Read page 104
NO2- As the 3 negative charge centres repel each other and take up positions in space to be as far apart as possible, the electron pairs are distributed in trigonal planar arrangement. With one lone pair of electrons, the actual structure of the ion is bent with a bond angle of about 1170 for O-N-O bond.
Molecules with more than one central atom Consider N2H4 , C2H2
Bond Polarity • Due to difference in electronegativity value between the 2 atoms in the bond. • Unequal distribution of electron density results in small charges on the atoms ( δ+ and δ- ) Example A dipole is established when two electrical charge of opposite sign are separated by a small distance. Dipole moment
Non-polar & polar covalent bonds Non-polar Covalent bond • No difference in electronegativity value – bond consists of 2 ____________ atoms. • _______ net charge. Examples : Polar Covalent bond • Due to the difference in electronegativity value – bond consists of 2 ____________ atoms. • _______ net charge. Examples:
Polar covalent compounds • Polarity of a molecule depends on the • the relative electronegativities of the atoms in the molecule and the shape of the molecule Which of the following is the most polar? HF, HCl, HBr or HI F, Cl, Br and I are in the same group (halogen). Electrnegativity decreases from F to I. Since bond polarity is due to the difference in electronegativity value, H-F is the most polar bond.
For CO2 each C-O bond is polar since O is more Electronegative than C. Why is the molecule non-polar?
There are some instances when the polar bonds are arranged symmetrically so as to give zero net direction of charge. i.e. Overall dipoles cancel so that there is no overall dipole. For example, carbon dioxide and carbon tetrachloride,