CHEMICAL INTERACTIONS
CHEMICAL INTERACTIONS. Chapter 1 - Chemical Reactions Outline/Review. 1.1 Matter and Its Changes. CHEMISTRY is the study of the properties of matter and how matter changes. An ELEMENT is a substance that cannot be broken down into any other substances by chemical or physical means.

CHEMICAL INTERACTIONS
E N D
Presentation Transcript
CHEMICALINTERACTIONS Chapter 1 - Chemical Reactions Outline/Review
1.1 Matter and Its Changes • CHEMISTRY is the study of the properties of matter and how matter changes. • An ELEMENT is a substance that cannot be broken down into any other substances by chemical or physical means. • All the matter around you is composed of one element or a combination of two or more elements.
A COMPOUND is a substance made of two or more elements chemically combined in a specific ratio. • An example of a compound is CO2 • A MIXTURE is made from two or more elements or compounds in the same place but not forming new material. • A SOLUTION is a well-mixed mixture where substances are not easily seen.
Chemistry is the study of the kinds of and changes in matter. • A PHYSICAL CHANGE is when the form or appearance of a material changes. • An example of a physical change is water in the forms of ice, water, and water vapor. • A CHEMICAL CHANGE is when a new substance is produced. • An example of a chemical change is metal rusting.
The change of font type is an example of a physical change. • Changing whatsomething says is an example of a chemical change. • You can observe a chemical reaction by observing changes in the properties of matter. • A PRECIPITATE is a solid formed during a chemical reaction.
Some of the evidence for chemical reactions is: • production of a gas • precipitation of a solid • a change in temperature (either up or down) • a color change • change in physical properties
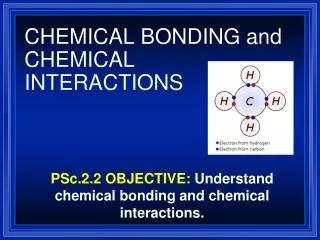
The smallest particle of an element is an ATOM. • A MOLECULE is the combination of two or more atoms. • An example of a molecule is H2O • An example of an atom is H • A CHEMICAL BOND is the force that holds atoms and molecules together. • Chemical reactions occur when chemical bonds are either formed or broken apart.
Therefore, to summarize . . . • Matter may be in the form of elements, compounds, or mixtures. • Chemical changes result in the formation of new substances. • Color change, production of a gas or a precipitate, a change in temperature, or a change in the properties of a substance are all clues that a chemical reaction has taken place. • Chemical reactions occur when chemical bonds are formed or broken
1.2 Describing Chemical Reactions • A CHEMICAL EQUATION is a shorter, easier way to show chemical reactions. • Most elements are represented by a 1-2 letter symbol. • An example of a symbol is O for oxygen. • A compound is represented by a “word”, called a CHEMICAL FORMULA. • An example of a chemical formula is H2O. • A SUBSCRIPT shows the number of atoms of an element in a molecule.
The materials you have at the beginning of a reaction are the REACTANTS. • The materials you have at the end of a reaction are called the PRODUCTS. • A chemical equation uses symbols to show the reactants and products of a chemical reaction. • An example of a chemical reaction is: H2 + O2 -----> H2O2 (H2 and O2 are the reactants while H2O2 is the product.)
All the atoms present at the start of a reaction are present at the end. • The law of conservation of mass states: The amount of matter in a chemical reaction does not change, so the total mass of the reactants must equal the total mass of the products. • A balanced chemical equation has the same amount of atoms for the reactants and the products. • A COEFFEICIENT can be added to balance the number of atoms in an equation.
An example of a balanced equation is: 2 Mg + O2 -----> 2 MgO • Many reactions can be classified in one of three categories: • SYNTHESIS • DECOMPOSITION • REPLACEMENT
A SYNTHESIS reaction occurs when two or more substances combine to make a more complex compound. • An example of a synthesis reaction is: 2 SO2 + O2 + 2 H2O -----> 2 H2SO4 • A DECOMPOSITION reaction breaks down compounds into simpler products. • An example of a decomposition reaction is: 2 H2O2 -----> 2 H2O + O2
A REPLACEMENT reaction is when one element replaces another in a compound OR when two elements trade places. • Examples of two replacement reactions are: 2 CuO + C -----> 2 Cu + CO2 MgCl2 + K2S -----> MgS + 2 KCl
Therefore, to summarize . . . • A chemical equation uses symbols to show the reactants and products of a chemical reaction. • Matter is neither created nor destroyed during a chemical reaction. • Chemical reactions may be classified by the types of changes in reactants and products.
1.3 Controlling Chemical Reactions • Every chemical reaction involves a change of energy. Some reactions release energy and others absorb energy. • An EXOTHERMIC reaction releases energy in the form of heat. • An ENDOTHERMIC reaction absorbs energy and feels cold. • ACTIVATION ENERGY is the amount of energy needed to get a reaction started.
The rate of a reaction isaffected by such factors as concentration, surface area, and temperature. • Increasing the CONCENTRATION is one way to increase the rate of a reaction. • Increasing the SURFACE AREA is another way of speeding up a reaction. • Increasing the TEMPERATURE is a third way to speed up a reaction. • A CATALYST is a material that lowers the activation energy needed to start the reaction.
A type of biological catalyst is an ENZYME. • Enzymes provide a surface for the reactions to take place. • An INHIBITOR is a material used to slow down a reaction - often to make it less dangerous.
Therefore, to summarize . . . • Every chemical reaction involves a change in energy. Some reactions absorb energy and others release it. • The rate of a chemical reaction can be controlled by such factors as concentration, surface area, temperature, and use of a catalyst or inhibitor.
1.4 Fire and Fire Safety • The rapid reaction between oxygen and a fuel is called COMBUSTION. • A FUEL is a material that releases energy when it burns. • Three things are necessary to start and maintain a fire--fuel, oxygen, and heat. • Water removes two parts of the fire triangle--heat and oxygen.
Some of the most common sources of fires are small heaters, kitchen fires, faulty electrical wiring, and carelessness with cigarettes (the most deadly). • Use baking soda to put out a fire that starts on the stove. • Try to extinguish small fires yourself, but leave a growing fire to the fire department. • Prevention is the best form of fire safety.
Therefore, to summarize . . . • The fire triangle shows the three things necessary to start a fire and keep it burning: fuel, oxygen, and heat. • Water stops combustion by keeping the fuel from coming in contact with oxygen. Also, evaporation of water uses a great deal of heat and cools the fire.
This concludes the Chapter 1 review for the Chemical Interactions book. STUDY WELL!