Measuring Solubility
Measuring Solubility. Chapter 11. Solubility. The solubility of a substance refers to the maximum amount of that substance that can be dissolved in a given quantity of solvent at a certain temperature.

Measuring Solubility
E N D
Presentation Transcript
Measuring Solubility Chapter 11
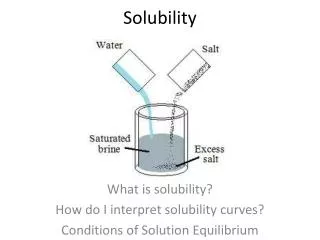
Solubility • The solubility of a substance refers to the maximum amount of that substance that can be dissolved in a given quantity of solvent at a certain temperature. • A solution in which no more solute can be dissolved at that temperature is described as a saturated solution.
Solubility • One way of measuring solubility is to determine the maximum mass of solute that can be dissolved in 100 grams of solvent at a particular temperature. • Solubility values allow us to compare the extent to which different substances dissolved. • Look at table 11.1 on page 207 and the worked example on the same page
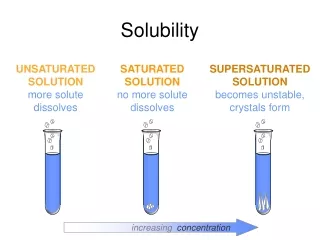
Solubility Curves • The relationship between solubility and temperature can be represented by a solubility curve. • Each point in the solubility curve represents a saturated solution. • Any point below a curve represents an unsaturated solution for that solute.
Example • An 80 g sample of NaNO3 is added to 200g of H2O at 20°C. Use the solubility curve in Figure 11.1 to calculate how much more NaNO3 needs to be added to make the solution saturated with NaNO3 at 20°C.
Crystallisation • You might have noticed that honey often crystallises if you keep it in the refrigerator. The sugar becomes less soluble as the honey cools. • The sugar that will no longer stay dissolved comes out of solution as crystals. • This process is known as crystallisation.
Supersaturation • With some substances it is possible to produce an unstable solution that contains more dissolved solute than in a saturated solution. • Such a solution is said to be supersaturated. • Any point above a solubility curve represents a supersaturated solution.
Solubility of gases • Gases such as oxygen an carbon dioxide are much less soluble in water than solutes such as NaCl and sugars. Why? • But oxygen and carbon dioxide are present in our oceans and waterways. • The solubility of a particular gas in a liquid depends on the temperature of the liquid and the pressure of the gas.
Temperature and Gas Solubility • Unlike most solids, gases become less soluble as the temperature increases. • When you heat water, small bubbles of air form and escape the water.
Pressure • Soft drinks contain carbon dioxide. • It is forced into the cans under high pressure to increase the amount that can be dissolved. When the can or bottle is opened the carbon dioxide can escape. • This is how drinks get flat after a certain time. As more and more carbon dioxide escapes.
Your Turn • Look at worked example 11.1d on page 210. • Page 211 • Question 1 • Question 3 • Question 5 • Question 7
Concentration of solutions • Before we begin I am just warning you that this is the return of the mole. • The mole will continue right through til the end of unit 4. • It is vital you understand the mole, if you are unsure of anything stop me and ask. • If you are unsure chances are someone else in the class is unsure too.
Concentration of Solutions • When talking concentrate think of cordial. • If I pour the same amount of cordial into two glasses but have different amounts of water their concentrations are the same even if their volumes are different. • Volume and concentration are two different things.
Concentration of Solutions • The concentration of a solution describes the relative amounts of solute and solvent present. • A solution in which the ratio of solute to solvent is high is said to be concentrated. • A solution in which the ratio of solute to solvent is low is said to be dilute.
Concentration of Solutions • Chemists use different measures of concentration depending on the particular situation. • Earlier, units of grams of solute per 100 grams of solvent were used to describe the concentration of a saturated solution. • Other ways of expressing concentration describe the amount of solute in a given amount of solution. • They vary only in units used to measure the amount of solute and the amount of solutions.
Concentration of Solutions • For chemists, the most commonly used units for concentration are: • Mass of solute per litre of solution • Amount, in mol, of solute per litre of solution. (does this one look familiar)
Mass of solute per litre of solution • This unit expressed concentration in terms of the mass of solute present in 1L of solution. • It is important you know how to convert metric units of volume.
mass of sulfate ions (mg) Concentration = volume of mineral water (L) A 250ml glass of orange-flavoured mineral water contains 4.0mg of sulfate ions. What is the concentration (in mg L-1) of sulfate ions in the mineral water? Solution: Remember 250 ml is 0.250 L 250/1000 4.0 mg Concentration = 0.250 L Concentration = 16 mg/L or 16 mg L-1
Other units • Other units commonly used to measure volume are the cubic centimetre (cm3), the cubic decimetre (dm3) and the cubic metre (m3). • Where 1 mL = 1cm3, 1 L = dm3 and 1 KL = 1 m3
Your Turn • Page 215 • Question 9
Amount, in mol, of solute per litre of solution • Expressing concentration in moles per litre of solution allows chemists to compare relative numbers of atoms, molecules or ions present in a given volume of solution. • The measure of concentration, known as molarity or molar concentration, is an important one for chemists.
Molarity (M) • Molarity is defined as the number of moles of solute particles per litre of solution. • A one molar (1 M) solution contains one mole of solute dissolved in each litre of solution. • A concentration of such a solution is said to be one mole per litre, 1 mol L-1 or 1M. • We use the term molarity to mean ‘concentration measure in moles per litre.
Molarity (M) • 1.0 L of a 1.0 M solution of ethanol contains 1 mol of C2H5OH • 1.0 L of a 1.0 M solution of sodium chloride contains 1 mol of NaCl • 2.0 L or a 0.5 mol solution of sodium chloride contains 1 mol of NaCl • 0.25 L of a 4.0 M solutions of ammonia contains 1 mol of NH3. • Each of these solutions contains 1 mole of the solutes dissolved in solution.
Amount, mol Concentration, mol L-1 or M The Equation • The amount fo solute is linked to the concentration (molarity) and volume of the solution by the relationship: n = c x V Volume, L
Equation n = cV Or n n c = V = V c
Unit converstion • The concentration units discussed here are g L-1 and mol L-1. • We must be able to convert from one unit to the other at times. • Since litres is common to both we are really just converting from grams to mole and vice versa. • How do we convert from grams to mole again?
Don’t forget molar mass • Both molarity and molar mass use M at times. Molarity uses it as units where as molar mass uses it as a symbol. Always look carefully to determine which one M means in each question. ÷ M grams moles litre litre x M
Worked examples • Lets do the worked examples together. • Page 213
Your Turn • Page 215 • Question 10, 11 and 12
Dilution • If I don’t like my cordial strong I add more water. • I am in effect adding more solvent (water) to a solution (cordial). • This process is known as dilution. • When a solute is diluted there is still the same amount of solute. The solute particles, however, are more widely spaced.
Dilution • Because the amount (number of moles) of solute does not change during dilution, a useful mathematical relationship exists. • Amount of solute before dilution is n1. n1 = c1V1. • Amount of solute after dilution is n2. n2 = c2V2. • But n1 = n2. • So: • c1V1 = c2V2.
Dilution c1V1 = c2V2 • This is called the dilution formula. • Since the amount of solute remains unchanged during dilution, so does the mass of the solute. Page 214 Worked examples.
Your Turn • Page 215 • Question 13