Solubility
Solubility. Definition. Q. How do you measure a compound’s solubility?. A. The amount of that compound that will dissolve in a set volume of water. This is usually measured in gL -1 or molL -1. What happens as a sparingly soluble solid dissolves?. E.g. Dissolving CuSO 4 in water.

Solubility
E N D
Presentation Transcript
Definition Q. How do you measure a compound’s solubility? A. The amount of that compound that will dissolve in a set volume of water. This is usually measured in gL-1 or molL-1.
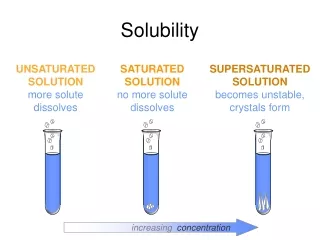
What happens as a sparingly soluble solid dissolves? E.g. Dissolving CuSO4 in water. As you add solid CuSO4, it dissolves until it reaches the saturation point. This is the point at which no more solid will dissolve. You know when you’ve reached saturation when____________________________. At this point an equilibrium system is established where the rates of the forward and reverse reactions are equal. Write an equation for this equilibrium reaction. CuSO4 (s) Cu2+ (aq) + SO42- (aq) Now write a K expression for this equilibrium. Ks = [Cu2+ (aq)] [SO42- (aq)] The Ks is the SOLUBILITY PRODUCT – the product of the concentration of ions in a saturated solution of a sparingly soluble compound.
Questions: Calculate the concentration of all the ions in a saturated solution of the following: 1. Calcium fluoride (solubility = 2.2 x 10-4 molL-1) 2. Copper (II) sulfide (solubility = 2.5 x 10-18 molL-1) 3. Calcium hydroxide (solubility = 1.1 x 10-2 molL-1) 4. Silver sulfide (solubility = 1.2 x 10-17 molL-1) 5. Silver iodide (solubility = 9.1 x 10-9 molL-1) Answers: 1. Ca2+ = 2.2 x 10-4 molL-1, F- = 4.4 x 10-4 molL-1 2. Cu2+ = 2.5 x 10-18 molL-1, S2- = 2.5 x 10 -18 molL-1 3. Ca2+ = 1.1 x 10-2 molL-1, OH- = 2.2 x 10-2 molL-1 4. Ag+ = 2.4 x 10-17 molL-1, S2- = 1.2 x 10-17 molL-1 5. Ag+ = 9.1 x 10-9 molL-1, I- = 9.1 x 10-9 molL-1
Calculating the solubility product (Ks) given the solubility 1.50 x 10-9 1. Calculate the Ks for BaSO4 given its solubility = 3.87 x 10-5 molL-1 2. Calculate the Ks for CaF2 given its solubility = 2.2 x 10-4 molL-1 3. Calculate the Ks for PbI2 given its solubility = 4.1 x 10-2 gL-1 4. Calculate the Ks for silver sulfide given its solubility = 1.2 x 10-17 molL-1 4.26 x 10-11 2.8 x 10-12 6.91 x 10-51 First decide if the compound is AB, AB2 or A2B type. Write an equilibrium equation and a Ks expression for the compound. Decide on the concentration of each ion using the solubility. Substitute the ion concentrations into the Ks expression and solve.
Write a general formula Try writing a general formula for calculating Ks from solubility. Let solubility = x. Your answer will be different for AB (e.g. CaSO4) compounds than AB2 (e.g. CaF2) and A2B (e.g. Ag2S) compounds. Answer: AB: Ks = x2 AB2 and A2B: Ks = 4x3
Remember: • The solubility has to be DOUBLED for A (in A2B compounds) and B (in AB2 compounds. This is because that ion is present at TWICE the concentration of the other ion, and equilibrium constants are a product of the CONCENTRATION of ions!
Going the other direction… Calculate the solubility of the following compounds given their Ks value: • The Ks for silver chloride is 1.7 x 10-10. Calculate its solubility. • The Ks for lead chloride is 1.7 x 10-5. Calculate its solubility. • The Ks for silver sulfate is 1.22 x 10-5. Calculate its solubility. • The Ks for calcium hydroxide is 1.0 x 10-2. Calculate the pH of a saturated solution of calcium hydroxide. • Answers: • 1.3 x 10-5 molL-1 (using Ks = x2) • 0.0162 molL-1 (using Ks = 4x3) • 0.0145 molL-1 (using Ks =4 x3) • 13.4 (using Ks = 4x3, then [OH-]=2 x solubility, then pH = 14—log[OH-])
Key points • The Ks is the SOLUBILITY PRODUCT. It refers to a saturated solution ONLY – i.e. dissolving a maximum amount of a solid in water • The solubility in molL-1 = X • X also equals the concentration of the single ion (A and B in AB compounds, A in AB2 compounds and B in A2B compounds)
Ionic product • To be useful, you have to be able to use solubility data to PREDICT PRECIPITATION. E.g. What if we added fluoride ions to our water supply – would anything precipitate out producing a nasty solid floater in our drinking water? • We need to use the IONIC PRODUCT.
Ionic product and Ks • The ionic product is the product of the ions in solution, raised to the appropriate power • It is calculated in the same way as Ks HOWEVER, THEY ARE DIFFERENT!!!!! • Ks is calculated when you make a saturated solution of a sparingly soluble compound only (i.e. Dissolve stuff in water to saturation) • IP is calculated from any source of ions
Example questions 1. Calculate the Ks of BaSO4, given that a saturated solution of BaSO4 contains 3.87 x 10-5 molL-1 of Ba ions. Answer: Ks = [Ba2+][SO42-], = x2 = (3.87 x 10-5)2 = 1.5 x 10-9 2. Calculate the IP of BaSO4 if 4.24 x 10-4 molL-1 Ba2+ ions is added to a water supply containing 2.21 x 10-6 molL-1 sulfate ions. Answer: IP = [Ba2+][SO42-], = (4.24 x 10-4)( 2.21x10-6) = 9.37x 10-10
Using IP From the previous page: Will a precipitate form in the water supply? Compare IP to Ks If IP > Ks, then concentration of ions exceeds solubility so a PRECIPITATE FORMS If IP < Ks, then concentration of ions is less than solubility so NO PRECIPITATE FORMS See Pearson power point “ionic product”
Common ion effect • The presence of a common ion decreases a compounds solubility e.g. KCl is less soluble in water than it is in an NaCl solution. This is because the salt solution contains one of the ions you are trying to dissolve KCl ↔ K+ + Cl- The presence of Cl- in the solution pushes the equilibrium to the left, thus precipitating KCl • See Pearson Powerpoint “common ion effect”
Common ion question • Calculate the solubility of PbI2 in pure water and in 0.1 molL-1 KI. Ks(PbI2) = 1x10-9. Explain your answer using equilibrium principles.
Complex ions Remember these? Ag(NH3)22+ Zn(NH3)42+ Pb(OH)42- Zn(OH)42- Al(OH)4- Cu(NH3)42+ Complex ions – made from a central metal ion and a number of ligands. These are soluble. The presence of one of these ligands can allow complex ions to form, which can INCREASE the solubility of some sparingly soluble compounds
Example • Suppose we have a saturated solution of AgBr, with some AgBr(s) left undissolved at the bottom of the beaker. We next add some aqueous ammonia to the system. Its molecules are strong ligands for silver ions, so they begin to form Ag(NH3)2+ ions from the trace amount of Ag+(aq) initially present in solution. • Ag+(aq) + 2 NH3(aq) <==> Ag(NH3)2+(aq) • The ammonia represents an upset to the equilibrium present in the saturated solution of • AgBr(s) <===> Ag+(aq) + Br-(aq) • By pulling Ag+ ions out of this equilibrium, the equilibrium must shift to the right to replace them as best it can. i.e. more AgBr(s) must go into solution. The solubility of a slightly soluble salt increases when one of its ions can be changed to a soluble complex ion.
The effect of added acid on basic anions • The pH also influences the solubility of salts that contain a basic anion, an anion from a weak acid for example. In general we would expect the salts of weak acids to be more soluble in acidic solutions. For example consider CaF2 • CaF2(s) <==> Ca2+(aq) + 2 F-(aq) • In an acidic solution the F- ions will be removed by reacting with H3O+ to form HF, thus shifting the equilibrium to the right and causing more CaF2 to dissolve. • H3O+ + F- <==> HF + H2O • The solubilities of salts containing anions that do not hydrolyze, such as Cl-, Br-, I-, NO3- in other words anions from strong acids are not affected by pH.