Bonding
Bonding. Writing Formula and Naming Compounds. STEPS. 1. Decide if compound is Ionic or Covalent: All Ionic Compounds must have a Cation (Positive Ion) and an Anion (Negative Ion) Covalent Compounds contain either Nonmetals or Metalloids. Steps For Ionic Compounds.


Bonding
E N D
Presentation Transcript
Bonding Writing Formula and Naming Compounds
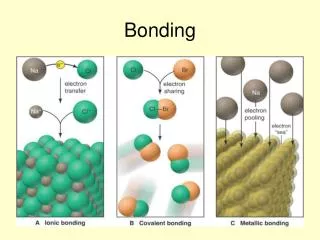
STEPS • 1. Decide if compound is Ionic or Covalent: • All Ionic Compounds must have a Cation (Positive Ion) and an Anion (Negative Ion) • Covalent Compounds contain either Nonmetals or Metalloids
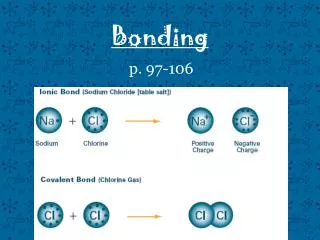
Steps For Ionic Compounds • 1. Write down the symbols of the ions in the compound • 2. Determine the charge on each ion and write the charges as superscripts • 3. Criss-cross the quantity of the charges (only the number not the sign) • 4. Write the quantity as subscripts • 5. Reduce (if necessary) subscripts to get lowest ratio
Example 1: Calcium Chloride • Calcium • Ca • Ca+2 • Chloride • Cl • Cl-1 CaCl2
Example 2: Iron (II) Phosphate • Iron (II) • Fe • Fe +2 • Phosphate • PO4 • PO4-3 Fe3(PO4)2 Note: Because ‘2’ phosphates are needed to equal the charge of 3 Fe +2, phosphate was placed inside ( ).
Covalent Compounds The following prefixes are used to indicate the number of atoms present in the compound for each element. Note: atoms do notget reduced as in Ionic Compounds. • Mono = 1 • Di = 2 • Tri = 3 • Tetra = 4 • Penta = 5 • Hexa = 6 • Septa = 7 • Octa = 8 • Nona = 9 • Dec = 10
Example 3: Carbon Tetrachloride(covalent since no metal is present) • Carbon • C • Chloride • Cl Since ‘tetra’ means 4, there are 4 atoms of Chlorine in the compound and the formula is: CCl4
Example 4: Dinitrogen Trioxide • Nitrogen • N • Oxygen • O Since “di’ means 2 and “tri’ means 3, the formula is: N2O3
Naming Ionic Compounds • Since ionic formulas are written by criss-crossing the charges, to name them just “uncriss-cross” the formula. • Remember, ionic formulas are the simplest ratio of the atoms present for each element. So you have to keep in mind if any quantities got reduced.
Example 5: Fe2(SO4)3 • Uncriss-crossing shows that the 3 came from the Fe (iron) and the 2 from the SO4 (sulfate) • Name of the compound is Iron (III) Sulfate
Example 6: CuO • Quickly looking at this formula, you may think there’s 1 atom of each element. BUT, remember, Oxygen gains 2 electrons and forms the O -2 anion. • This means that the charge from the Copper ion must balance the charge of the Oxygen ion. • So, the correct name of this compound is: • Copper (II) Oxide
Naming Covalent Compounds • Look at the number of atoms of each element present and use the prefixes to name the compound • If the first element has only 1 atom, do not use ‘mono’ • If using the prefix as is makes the name sound ‘weird’, just drop the vowel: CO = Carbon Monoxide and not Carbon Monooxide
Example 7: P4S5 • P = Phosphorus • S = Sulfur • Tetra = 4 • Penta = 5 • TetraphosphorusPentasulfide