Understanding the Central Dogma of Molecular Biology: DNA, RNA, and Protein Synthesis
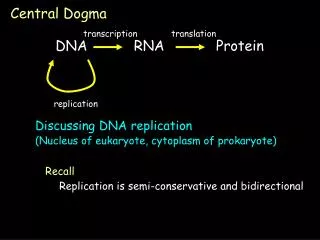
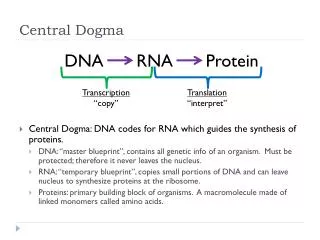
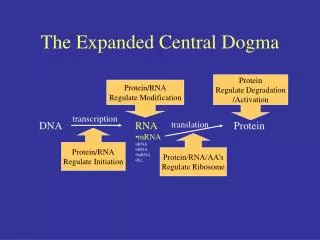
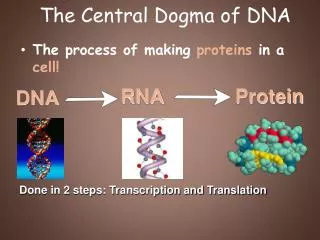
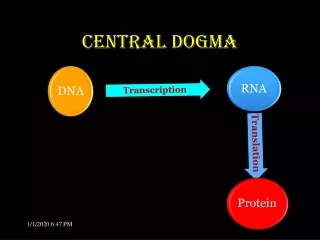
The central dogma of molecular biology elucidates the flow of genetic information within a biological system. DNA replication produces identical copies, while transcription generates RNA from DNA, leading to protein synthesis through translation. This process determines cellular functions based on the types and amounts of proteins expressed. The structure of DNA, composed of nucleotides with a stable backbone, facilitates efficient information storage and retrieval. Understanding these mechanisms is crucial for advancements in genetics, biotechnology, and medicine.

Understanding the Central Dogma of Molecular Biology: DNA, RNA, and Protein Synthesis
E N D
Presentation Transcript
The Dogma • Nucleic acid (DNA/RNA) is important…Why? • The central dogma of Molecular Biology • DNA (genes, chromosomes) begets itself (replication), as well as RNA (transcription)) • RNA begets protein (translation) • Which proteins a cell expresses (and how much), dictates what a cell does
DNA structure • DNA: an ideal molecule for storage of information. • Made of simple, stable(?) “bits” of information (the nucleotide) (metaphor: letters) • Easily assembled/disassembled (metabolism) (metaphor: words, sentences, books) • The information is easily “read” (replication, transcription)
The nucleotide: Pentose sugar 4’ 1’ Pentose Sugar (2’ OH=ribose, 2’H=deoxyribose)
The nucleotide: Nitrogenous bases Pyrimidines (small) Purines (BIG) From Kimball’s biology pages: http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/N/Nucleotides.html
The nucleotide Phosphate Base Sugar (2’ OH=ribose, 2’H=deoxyribose) Nucleotide =sugar+phosphate+base
The chain has polarity DNA chains are connected by a phosphate between sugar carbons The chain has polarity: the phosphate bridges between 5’ and 3’ carbons (almost never 5’ and 5’, or 3’ and 3’)
DNA metabolism • Making phosphodiester bonds… • Synthesis (Nucleotide addition, nucleotide by nucleotide) • Ligation (joining two polynucleotide chains together) • Breaking phosphodiester bonds…. • Cleavage, or hydrolysis
Synthesis Chemistry dictates addition is always to 3’ end of chain. In other words: synthesis is always 5’ to 3’
DNA synthesis • Synthesis: requires... • Substrates • 3’ OH of existing chain (primer strand) • template strand (see replication lecture) • deoxynucleotide triphosphate (dNTP) • Cofactors • Mg2+ (metal cofactor) • Enzyme (DNA polymerase) • Products are… • Chain that is longer by one nucleotide • Pyrophosphate (PPi)
Cleavage (hydrolysis) • Chain is broken between phosphate and sugar (5’ carbon usually retains phosphate) • Requires…. • Substrate: • DNA chain, usually double stranded • Water • Enzyme (nuclease) • Co-factors; usually Mg2+ • Product: broken chain • If chain broken from end, enzyme is exonuclease • Exonucleases can chew from 3’ end (3’ to 5’ exo) or 5’ end (5’ to 3’ exo) • If chain broken in middle, enzyme is endonuclease
Cleavage: Endonuclease Restriction enzymes are endonucleases (see Lee lecture)
Ligation • Requires… • Substrates • two DNA chains • ATP • Cofactors • Mg2+ (metal cofactor) • Enzyme: ligase • Products are… • Two chains joined together into one chain • AMP • Pyrophosphate (PPi)
DNA chains form helices • Single DNA chains will form a helix (spiraling line; like threads on screw) because of…. • Hydrophobic interactions between bases • Bases are carbon rich rings that hide from water, and therefore stack on top of each other • Ionic interactions • Phosphates are highly negatively charged, thus repel each other
The double helix • A single stranded DNA chain will form a helix but… • Each base has a number of hydrogen donors and acceptors • Donors like to form hydrogen bonds with acceptors • Like this…..
Watson-Crick base pairs • A with T • G pairs with C • Why? • Complementary pattern of hydrogen donors and acceptors • GC stronger than AT • From http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/B/BasePairing.html
The double helix • Two chains with extended sequence that can pair together, or is “complementary”, may form a double helix • Constraints of backbone structure permits double helices only when complementary sequence is of the opposite polarity. • i.e….5’GGTCA3’ will pair with 5’TGACC3’, but NOT 5’CCAGT3’
Association/disassociation of the double helix • Hydrogen bonds between paired bases are weak • Sensitive to temperature, salt concentration • Heating will separate, denature, or melt a double helix into two separate stands (single stranded, or ssDNA) • Denaturation occurs at a specific temperature (melting temperature, or Tm) • Tm defined by length (longer comlementary sequence=higher Tm) and sequence (higher GC%=higher Tm) • Separation of strands required for replication, transcription • instead of heat, these processes use ATP for energy to break base pairs
Secondary structure Secondary structure hairpins (intra-molecular pairing of single strand) heteroduplex (double stranded DNA with the occasional mismatch, forms “bubbles” in double helix. Can be caused by renaturation of partially complementary sequence, or replication errors
Helical shape • Helical parameters… • Screw sense: left handed, or right handed • Twist: degrees rotation, along the horizontal axis, between successive base pairs • Rise: elevation, along the vertical axis, between successive base pairs • Tilt: degrees of inclination of base pair from the horizontal access (in most double helices base pairs are not significantly tilted )
Helical forms • A form • Formed in DNA under dehydrating conditions • Major form of RNA double helix • B form • Standard DNA double helix • Z DNA • Forms in vitro primarily at GC rich regions
Helical forms • A form • Shorter, fatter than B form DNA • High degree of base pair tilt • B form • Standard DNA double helix • About 34o twist, 3.4 angstrom rise, very little tilt • Z DNA • Only left handed helix • Kinked backbone (does not smoothly conform to helical shape) • Much greater rise, reduced twist relative to B DNA