Chromatography
Chromatography. Retention Factor (RF) Thin Later Chromatography (TLC) Gel Filtration Chromatography Ion Exchange Chromatography. Chromatography is the method used by scientists for separating organic and inorganic compounds so that they can be analyzed and studied.

Chromatography
E N D
Presentation Transcript
Chromatography Retention Factor (RF) Thin Later Chromatography (TLC) Gel Filtration Chromatography Ion Exchange Chromatography
Chromatography is the method used by scientists for separating organic and inorganic compounds so that they can be analyzed and studied. • Chromatography means (chromo= colour & graphy= to write). • Russian botanist M.S. Tswett invented chromatography in 1903. • Over 60% chemical analysis worldwide is currently done with the help of chromatography. • Chromatography is used in many fields such as to determine unknown substances, police use it for solving criminal cases, to determine the presence of cocaine in urine, alcohol in blood and lead in water.
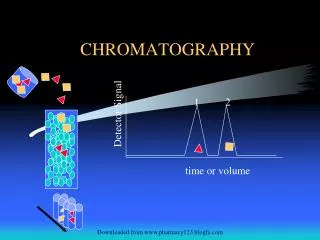
Chromatography is based on differential migration. • In all chromatography there is a mobile phase and a stationary phase. • The solutes in a mobile phase go through a stationary phase. • The solutes move through the stationary phase and gets separated, this is chromatographic development. • In paper & thin layer chromatography the mobile phase is the solvent. • The stationary phase in paper chromatography is a strip of piece of paper placed in a solvent & in (TCL) it is thin layer of solid as silica supported on inert base such as glass.
Retention Factor: • Retention factor/ ratio (Rf) is a quantitative indication of how far a particular compound travels in a particular solvent. • The Rf value is a good indicator of whether an unknown compound and known compounds are similar, if not identical. • The retention factor/ratio is defined as a distance traveled by solute(x) divided by distance traveled by the solvent (y). • Retention ratio (Rf) distance moved by solute distance moved by solvent
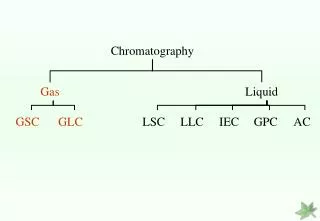
There are several types of chromatography currently in use… • Paper chromatography • Thin layer chromatography • Gas chromatography • Liquid chromatography • High performance liquid chromatography (HPLC) • Gel filtration chromatography • Ion exchange chromatography
Thin layer chromatography (TLC): • In thin layer chromatography the stationary phase is a thin layer of a solid such as alumina or silica gel or cellulose powder, supported on an inert base such as glass, aluminum foil or insoluble plastic. • The thin layer is allowed to dry at room temperature and is then activated by heating in as oven between 100 -250 degrees. • With the help of micropipette the mixture is carefully spotted at the bottom of TLC plate. • The plate is placed in a closed vessel containing solvent so that the liquid level is below the spot.
Applications: • It can be used in organic , inorganic chemistry, bio-chemistry & pharmaceutical labs. • Used for the separation and identification of components of mixtures on very small scale and very quickly. • It gives more reproducible Rf value. • It can be used for adsorption, partition, ion exchange types of chromatography. • It makes the use of radioactive isotopes successfully. • In forensic science, TLC is used to detect poisons, metals, drugs, tranquilizers and other substances in body fluids.
Gel Filtration Chromatography: • Gel filtration chromatography is also known as size exclusion chromatography or molecular sieve chromatography or permeation chromatography. • In this technique, molecules are separated on the basis of their size & shape. • The sample in a small volume is applied to the top of a column of porous beads (diameter 0.1 mm) that are made of an insoluble but highly hydrated spongy material such as polyacrylamide (Bio-gel) . • Small molecules can enter the pores in the beads whereas larger or more elongated molecules cannot. • The larger molecules will pass through inter-bead space, they will freely pass down the column more rapidly.
The larger molecules emerging out of the bottom (eluting) first & smaller molecules move slowly through the column and elute later. • Beads of different pore sizes are used to separate proteins effectively. • Application: • 1. Purification of biological macromolecules. • 2. Determination of relative molecular mass studies. • 3. Solution of substances with high molecular mass can be concentrated by using swelling gel. • 4. This technique is useful in protein binding studies.
Fig: showing separation of smaller and larger molecule in Gel filtration
Ion Exchange Chromatography • Ion exchange chromatography is an adsorption chromatography. • In this type, the solid stationary phase is an ion exchange resin and mixture is in the mobile phase. • The separation of ions or polar compounds present in the sample and transportation by the mobile phase is result of their interaction with ionic sites on the stationary phase. • The retention of a compound in a stationary phase depends on its charge density. • Higher the charge density, longer the compound will retained on the stationary phase.
Depending on the compounds of the mixture to be separated the cation or anion exchange resin is selected. • The column is filled with ion exchange resin selected. The mixture is the form of solution is poured from top and allowed to drain down. • Then eluting solution is added and allowed to drain down. • Columns containing positively charged diethylaminoethyl (DEAE) groups such as DEAE- cellulose or DEAE- sephadex) are used for separation of negatively charged proteins (anionic proteins) . • This is called anion exchange chromatography.
Columns containing negatively charged carboxymethyl (CM) groups (such as CM- cellulose or CM –sephadex) are used for the seperation of positively charged proteins (cationic proteins). • This is called as cation exchange chromatography. • Applications: • Purification of water • Extraction of Metals from solution • Separation of Amino acids