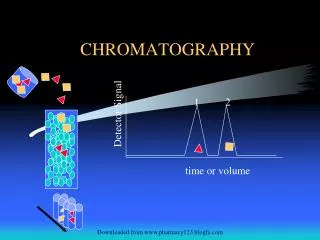
Chromatography
Chromatography. What is it??. It is Greek…. " chroma " meaning color and " graphein " to write. Uses Of Chromatography. Separating mixtures to get pure substances Biotechnology Can purify any volatile substances under the right conditions Can separate proteins Separate delicate substances

Chromatography
E N D
Presentation Transcript
Chromatography What is it??
It is Greek… "chroma" meaning color and "graphein" to write
Uses Of Chromatography • Separating mixtures to get pure substances • Biotechnology • Can purify any volatile substances under the right conditions • Can separate proteins • Separate delicate substances • Forensics • Athletics • Isolating strands of DNA
Steps… 1. Prepare the candy by placing it on the aluminum foil in a drop of water. Turn the candy after a minute to ensure that all the color is off of the candy.
Step 2… Prepare the coffee filter by cutting out a 3x3 square and labeling the letters of the colors of the skittles 1/2inch from the bottom. Make sure you are 1/4inch from both sides of the filter as well (see picture)
Step 3… Dip a tooth pick into your first colored drop of water that you created in Step 1, and make a small dot of color on your coffee filter where you made your first mark. It is best to keep these dots as small as possible. Let the liquid dry, and then repeat this step twice more to get a lot of pigment on your filter paper.
Step 4… • Pour the salt water solution into a beaker (I have done this for you)Place your coffee filter paper in the cup (Only after it has dried)-- the water should only touch the bottom, and not reach the colored marks you made. If there is too much, dump some of the salt solution out.
Step 5…Watch the Capillary Action! • As the salted water travels up the paper through capillary action, it will carry some pigment along with it. Different pigments have different affinities for the salt water, causing them to travel varying distances up the filter paper.
Rf Values… • In order to make the technique more scientific rather than a mere interpretation by sight, what is called the Retention Value (Rf value for short) was applied in chromatography. A particular compound will travel the same distance along the stationary phase by a specific solvent (or solvent mixture) given that other experimental conditions are kept constant. In other words, every compound (dye, pigment, organic substance etc) have a specific Rf value for every specific solvent and solvent concentration. Rf values come very handy for identification because one can compare Rf values of the unknown sample (or its consituents) with Rf Values of known compounds.