Atoms and Molecules
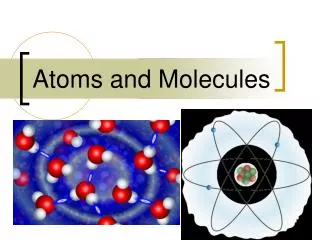
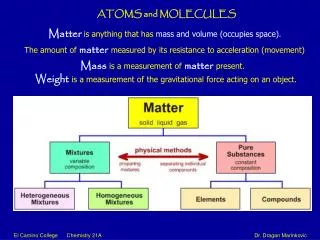
Atoms and Molecules. Matter is made of atoms. Brainpop -Atoms. Models. In the case of atoms, scientists use large models to explain something that is very small . Atoms are so small, it would take about 1,000,000 lined up in a row to equal the thickness of a human hair.

Atoms and Molecules
E N D
Presentation Transcript
Matter is made of atoms Brainpop-Atoms
Models • In the case of atoms, scientists use large models to explain something that is very small . Atoms are so small, it would take about 1,000,000 lined up in a row to equal the thickness of a human hair. • Models of the atom were used to explain data or facts that were gathered experimentally.
Atomic Structure Discovery Education Video Clip-Click Here
Click Here to watch a fun song of all the elements Atoms of the same element are all alike, but are different from atoms of other elements.
Molecule 2 or more atoms join together chemically Molecules are groups of atoms
Water is a molecule. It’s chemical formula is H2O. That means that one water molecule is made up of 2 hydrogen atoms and one oxygen atom.
Here is a model of a molecule. It is sugar. C6H12O6 Click Here to hear the MEET THE ELEMENTS song
Heat • When molecules of any kind of atoms get more energy in them than they had before, they move faster, and we call that "heat". When the molecules move faster, they hit against each other and bounce apart, and so they end up further apart from each other than they were before. • Things are hot if their molecules are moving quickly, and cold if their molecules are moving more slowly. Temperature is a way of measuring how fast the molecules are moving. Brainpop-temperature Click Here to watch the Ball and Ring Heat Demonstration
Melting To melt means to change a substance from a solid to a liquid state by heating it to the melting point. Different substances melt at different temperatures. Water melts at 0°C or 32°F.
Rock melts at about 625 degrees Celsius Chocolate melts at about 34 degrees Celsius
Freezing To freeze means to change a substance from a liquid to a solid state by cooling it to the freezing point.
Boiling Point The boiling point is the temperature at which a liquid changes to a vapor or gas. Again this is a different temperature for different substances. For water the boiling point is 100°C or 212°F.
What is the boiling point of rubbing alcohol? • Pour 100 mL of isopropyl alcohol (C3H8O) into a beaker. • Put in a thermometer • Heat until boiling and read the temperature It should be about 84o