Atoms and molecules
Atoms and molecules. By: Sabrina Karasik & Kyla Formey. Atoms. Atoms are the basic building blocks of matter that make up everyday objects. A desk, the air, even you are made up of atoms!

Atoms and molecules
E N D
Presentation Transcript
Atoms and molecules By: Sabrina Karasik & Kyla Formey
Atoms • Atoms are the basic building blocks of matter that make up everyday objects. A desk, the air, even you are made up of atoms! • There are 90 naturally occurring kinds of atoms. Scientists in labs have been able to make about 25 more. • They are made with 3 basic particles: • Protons • Neutrons • Electrons
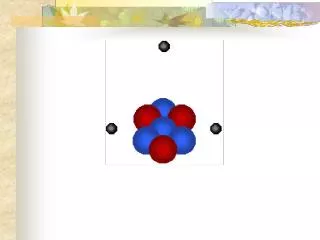
Atoms • An electron is negatively charged • A proton is positively charged • A neutron is neutral (no charge) • Adding a proton forms a new kind of atom • The protons and neutrons cluster together in the central part of the atom, called the nucleus and the electrons 'orbit' the nucleus. • A particular atom will have the same number of protons and electrons and most atoms have at least as many neutrons as protons.
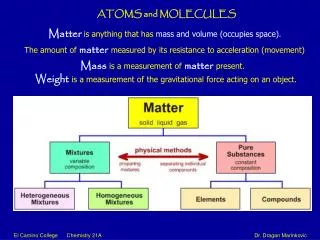
Molecules • A molecule is formed when two or more atoms are joined together by chemical bonds. • All compounds are molecules but not all molecules are compounds. • A compound is a molecule that is made up of at least 2 different elements. • Some examples of molecules are H2O (water) , N2 (nitrogen) , O3 (ozone) , CaO (calcium oxide) , • C6 H12 O6 (glucose)
Molecules • Molecules come in different shapes, sizes and complexities. • Molecules are always in motion. • They are packed tightly together in solids and liquids. • In a solid molecules have rapid vibration. • In a liquid molecules move around freely among each other.
Molecules • In a gas molecules are able to move more freely because the density of the molecules are less than in a solid and a liquid. • The speed of molecular motion increases as the absolute temperature increases. • Single atoms of elements such as a single O (oxygen) are not molecules. But if that oxygen bonds with itself or another element then it forms a molecule.