2 pt
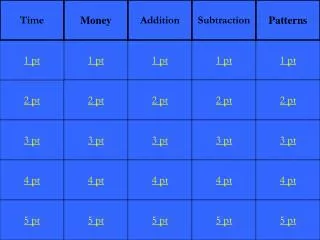
Ch. 1 and Chemistry. Prop of H20 Funct groups . Organic Compounds. Parts of The cell. Cell Membrane. 1pt. 1 pt. 1 pt. 1pt. 1 pt. 2 pt. 2 pt. 2pt. 2pt. 2 pt. 3 pt. 3 pt. 3 pt. 3 pt. 3 pt. 4 pt. 4 pt. 4pt. 4 pt. 4pt. 5pt. 5 pt. 5 pt. 5 pt. 5 pt.

2 pt
E N D
Presentation Transcript
Ch. 1 and Chemistry Prop of H20 Funct groups Organic Compounds Parts of The cell Cell Membrane 1pt 1 pt 1 pt 1pt 1 pt 2 pt 2 pt 2pt 2pt 2 pt 3 pt 3 pt 3 pt 3 pt 3 pt 4 pt 4 pt 4pt 4 pt 4pt 5pt 5 pt 5 pt 5 pt 5 pt
A localized group of organisms that belong to the same species is called a A) population. B) community. C) biosystem. D) ecosystem. E) family.
Which of the following types of cells utilize deoxyribonucleic acid (DNA) as their genetic material but do not have their DNA encased within a nuclear envelope? A) animal B) plant C) archaea D) fungi E) protists
Organisms interact with their environments, exchanging matter and energy. For example, plant chloroplasts convert the energy of sunlight into A) the energy of motion. B) carbon dioxide and water. C) oxygen. D) kinetic energy. E) the potential energy of chemical bonds.
Each element is unique and different from other elements because of the number of protons in the nuclei of its atoms. Which of the following indicates the number of protons in an atom's nucleus? A) atomic mass B) atomic weight C) atomic number D) mass weight E) mass number
The ionic bond of sodium chloride is formed when A) chlorine gains an electron from sodium. B) sodium and chlorine share an electron pair. C) sodium and chlorine both lose electrons from their outer valence shells. D) sodium gains an electron from chlorine. E) chlorine gains a proton from sodium.
The slight negative charge at one end of one water molecule is attracted to the slight positive charge of another water molecule. What is this attraction called? A) a covalent bond B) a hydrophilic bond C) an ionic bond D) a hydrogen bond E) a hydrophobic bond
Which of the following effects is produced by the high surface tension of water? A) Lakes don't freeze solid in winter, despite low temperatures. B) A water strider can walk across the surface of a small pond. C) Organisms resist temperature changes, although they give off heat due to chemical reactions. D) Water can act as a solvent. E) The pH of water remains exactly neutral.
B) A water strider can walk across the surface of a small pond.
What is the pH of a solution with a hydroxyl ion [OH-] concentration of 10-10 M? A) pH 2 B) pH 4 C) pH 10 D) pH 12 E) pH 14
Which is the best description of a carbonyl group? A) an oxygen joined to a carbon by a single covalent bond B) a nitrogen and two hydrogens joined to a carbon by covalent bonds C) a carbon joined to two hydrogens by single covalent bonds D) a sulfur and a hydrogen joined to a carbon by covalent bonds E) a carbon atom joined to an oxygen by a double covalent bond
E) a carbon atom joined to an oxygen by a double covalent bond
A compound contains hydroxyl groups as its predominant functional group. Which of the following statements is true concerning this compound? A) It lacks an asymmetric carbon, and it is probably a fat or lipid. B) It should dissolve in water. C) It should dissolve in a nonpolar solvent. D) It won't form hydrogen bonds with water. E) It is hydrophobic.
Testosterone and estradiol (estrogen) are A) nucleic acids. B) carbohydrates. C) proteins. D) phospholipids. E) steroids.
What is the chemical mechanism by which cells make polymers from monomers? A) phosphodiester linkages B) dehydration reactions C) ionic bonding of monomers D) hydrolysis E) the formation of disulfide bridges between monomers
What is the structure shown in Figure 5.4? A) starch molecule B) protein molecule C) steroid molecule D) cellulose molecule E) phospholipid molecule
What maintains the secondary structure of a protein? A) ionic bonds B) peptide bonds C) hydrogen bonds D) disulfide bonds E) phosphodiester bonds
Which of the following is true of both starch and cellulose? A) They are both polymers of glucose. B) They are geometric isomers of each other. C) They can both be digested by humans. D) They are both used for energy storage in plants. E) They are both structural components of the plant cell wall.
A primary objective of cell fractionation is to A) view the structure of cell membranes. B) identify the enzymes outside the organelles. C) determine the size of various organelles. D) separate the major organelles so that their particular functions can be determined. E) crack the cell wall so the cytoplasmic contents can be released.
D) separate the major organelles so that their particular functions can be determined.
All of the following are part of a prokaryotic cell except A) DNA. B) a plasma membrane. C) ribosomes. D) mitochondria E) a cell wall.
Which type of organelle is primarily involved in the synthesis of oils, phospholipids, and steroids? A) ribosome B) lysosome C) smooth endoplasmic reticulum D) mitochondrion E) contractile vacuole
Grana, thylakoids, and stroma are all components found in A) vacuoles. B) chloroplasts. C) mitochondria. D) lysosomes. E) nuclei.
The differences among the three categories of cytoskeletal elements would suggest that each of the following has specialized roles. Which of the following is a correct match? A) microfilaments and chromosome movement B) microfilaments and cleavage furrow formation C) microfilaments and ciliary motion D) intermediate filaments and cytoplasmic streaming E) microfilaments and the nuclear lamina
Who proposed that the membrane is a mosaic of protein molecules bobbing in a fluid bilayer of phospholipids? A) H. Davson and J. Danielli B) I. Langmuir C) C. Overton D) S. Singer and G. Nicolson E) E. Gorter and F. Grendel
According to the fluid mosaic model of cell membranes, which of the following is a true statement about membrane phospholipids? A) They cannot move laterally along the plane of the membrane. B) They rarely flip-flop from one side of the membrane to the other. C) They occur in an uninterrupted bilayer, with membrane proteins restricted to the surface of the membrane. D) They are free to depart from the membrane and dissolve in the surrounding solution. E) They have hydrophilic tails in the interior of the membrane.
B) They rarely flip-flop from one side of the membrane to the other.
Cell membranes are asymmetrical. Which of the following is a most likely explanation? A) The cell membrane forms a border between one cell and another in tightly packed tissues such as epithelium. B) Cell membranes communicate signals from one organism to another. C) Cell membrane proteins are determined as the membrane is being packaged in the ER and Golgi. D) The "innerness" and "outerness" of membrane surfaces are predetermined by genes. E) Proteins can only span cell membranes if they are hydrophobic.
C) Cell membrane proteins are determined as the membrane is being packaged in the ER and Golgi.
Celery stalks that are immersed in fresh water for several hours become stiff and hard. Similar stalks left in a salt solution become limp and soft. From this we can deduce that the cells of the celery stalks are A) hypotonic to both fresh water and the salt solution. B) hypertonic to both fresh water and the salt solution. C) hypotonic to fresh water but hypertonic to the salt solution. D) hypertonic to fresh water but hypotonic to the salt solution. E) isotonic with fresh water but hypotonic to the salt solution.
D) hypertonic to fresh water but hypotonic to the salt solution.
Five dialysis bags, constructed from a semi-permeable membrane that is impermeable to sucrose, were filled with various concentrations of sucrose and then placed in separate beakers containing an initial concentration of 0.6 M sucrose solution. At 10-minute intervals, the bags were massed (weighed) and the percent change in mass of each bag was graphed. Which line represents the bag with the lowest initial concentration of sucrose?