Phospholipid structure
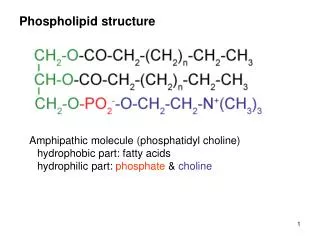
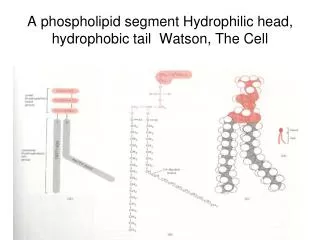
Phospholipid structure. Amphipathic molecule (phosphatidyl choline) hydrophobic part: fatty acids hydrophilic part: phosphate & choline. Membrane structure. Membrane components. Transmembrane proteins. Intercellular structures. Desmosomes

Phospholipid structure
E N D
Presentation Transcript
Phospholipid structure Amphipathic molecule (phosphatidyl choline) hydrophobic part: fatty acids hydrophilic part: phosphate & choline
Intercellular structures Desmosomes “spot welds”, dense proteins (cytoplasm & intercellular) fibers (intermediate filaments) extend across cells epithelial cells (especially skin), cardiac intercalated disks Tight junctions cell “collar”, block large molecules, no lateral protein movement epithelial cells Gap junctions cell-cell communication, small molecules (<1000 MWt) cardiac intercalated disks, smooth muscle
Desmosomes “spot welds”, dense proteins (cytoplasm & intercellular) fibers (intermediate filaments) extend across cells epithelial cells (especially skin), cardiac intercalated disks fig 3-10a
Tight junctions cell “collar”, block large molecules, no lateral protein movement epithelial tissue (esp. kidney, gut) paracellular pathway between cells fig 3-10b
Gap junctions cell-cell communication, small molecules (<1000 MWt) cardiac intercalated disks, smooth muscle fig 3-10d
Epithelial cell fig 3-10c
Protein ligand interaction Proteins could be: Ligands would be: enzymes substrates, allosteric regulators receptors chemical messengers transporters transported substances transcription factors transcription regulators any of above drugs
Protein-ligand binding properties Specificity: binding depends on ligand size, shape, charge Affinity: strength of binding: i.e. [ligand] at 50% binding Saturation: there is a finite number of binding sites Competition: structurally similar molecules can compete for binding
Specificity binding depends on ligand size, shape, charge fig 3-27 fig 3-26
Specificity protein Y specificity greater than protein X specificity fig 3-28
Affinity strength of binding: i.e. [ligand] at 50% binding fig 3-29
Affinity & saturation strength of binding: i.e. [ligand] at 50% binding fig 3-30
Affinity (different proteins) strength of binding: i.e. [ligand] at 50% binding fig 3-31
Affinity (different ligands) strength of binding: i.e. [ligand] at 50% binding
Protein-ligand binding properties Specificity: binding depends on ligand size, shape, charge Affinity: strength of binding: i.e. [ligand] at 50% binding Saturation: there is a finite number of binding sites Competition: structurally similar molecules can compete for binding and remember: the protein can be an enzyme, receptor, transporter, etc.
Regulating binding site properties a. Allosteric modulation reversible binding at another (“allo-”) site can be activation or inhibition
Regulating binding site properties a. Covalent modulation chemical alteration of the protein can be activation or inhibition
Metabolism (key) Key: A: glycogenesis, B: glycogenolysis, C: glycolysis, C+D: anaerobic glycolysis (lactic acid fermentation), E: gluconeogenesis, F: irreversible step (pyruvate dehydrogenase), G: protein synthesis, H: proteolysis, I: lipogenesis, J: lipolysis, K: Krebs cycle, L: urea synthesis, M: ketogenesis Anabolic pathways: A, G, I Catabolic pathways: B, C, E, F, H, J, K Liver only: E, L, M Mitochondrial: K Ribosomal: G Smooth endoplasmic reticulum: I