HEAT PPCI
HEAT PPCI. H ow E ffective are A ntithrombotic T herapies in PPCI. Heparin versus Bivalirudin in PPCI. Dr Adeel Shahzad Dr Rod Stables (PI) Liverpool Heart and Chest Hospital Liverpool, UK. Background. Anti-thrombotic therapy in PPCI

HEAT PPCI
E N D
Presentation Transcript
HEAT PPCI How Effective are Antithrombotic Therapies in PPCI Heparin versus Bivalirudin in PPCI Dr Adeel Shahzad Dr Rod Stables (PI) Liverpool Heart and Chest Hospital Liverpool, UK
Background • Anti-thrombotic therapy in PPCI • Selective (‘bailout’) use of GP IIb/IIIa antagonists (GPI) • Increasingly the norm in routine practice • Recommended by international guidelines • ESC ACCF / AHA
Background • Anti-thrombotic therapy in PPCI • Selective (‘bailout’) use of GP IIb/IIIa antagonists (GPI) • Increasingly the norm in routine practice • Recommended by key guidelines (ESC, ACCF / AHA) • Bivalirudin + selective (7% - 15%) use of GPI • Established anti-thrombotic treatment option
Background • Bleeding is associated with less favourable outcomes • Increased GPI use - results in increased bleeding • Observed for both bivalirudin and heparin • Relative performance of bivalirudin and heparin - • Cannot be reliably assessed with differential GPI use • HEAT PPCI • Bivalirudin + selective GPI v Heparin + selective GPI
Background • Bleeding is associated with less favourable outcomes • Increased GPI use - results in increased bleeding • Observed for both bivalirudin and heparin • Relative performance of bivalirudin and heparin - • Cannot be assessed reliably with differential GPI use • HEAT PPCI • Bivalirudin + ‘bailout’ GPI v Heparin + ‘bailout’ GPI
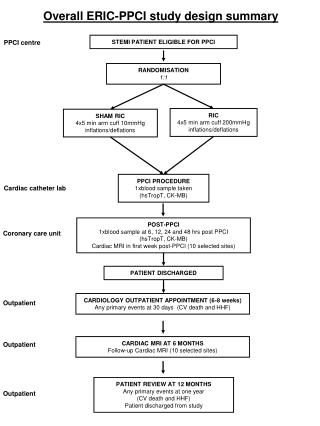
Study Description • Single centre RCT • Trial recruitment: Feb 2012 - Nov 2013 22 months • Bivalirudin v Unfractionated Heparin • STEMIpatients • Randomised at presentation • Acute phase management with Primary PCI
Study Description • Single centre RCT • Trial recruitment: Feb 2012 - Nov 2013 22 months • Bivalirudin v Unfractionated Heparin • STEMIpatients • Randomised at presentation • Acute phase management with Primary PCI • Philosophy for clinical teams: • Assess ‘Every Patient - Every Time’
Study Population Inclusion Criterion • All STEMI patients activating PPCI pathway Exclusion Criteria • Active bleeding at presentation • Factors precluding administration of oral A-P therapy • Known intolerance / contraindication to trial medication • Previous enrolment in this trial
Study Medication • Dual oral anti-platelet therapy pre-procedure • Heparin: 70 units/kg body weight pre-procedure • Bivalirudin: Bolus 0.75 mg/kg Infusion 1.75 mg/kg/hr - procedure duration
Study Medication • Dual oral anti-platelet therapy pre-procedure • Heparin: 70 units/kg body weight pre-procedure • Bivalirudin: Bolus 0.75 mg/kg Infusion 1.75 mg/kg/hr - procedure duration • GPI - Abciximab • Selective (‘bailout’) use in both groups • ESC guideline indications
Outcome Measures At 28 days Primary Efficacy Outcome Measure • Major Adverse Cardiac Events (MACE) - • All-cause mortality • Cerebrovascular accident (CVA) • Re-infarction • Unplanned target lesion revascularisation (TLR)
Outcome Measures At 28 days Primary Efficacy Outcome Measure • Major Adverse Cardiac Events (MACE) Primary Safety Outcome Measure • Major bleeding - • Type 3-5 bleeding as per BARC definitions
Study Organisation • Data Monitoring and Safety Committee (DMSC) • All key clinical events adjudicated • Clinical Events Committee • Blinded to the treatment allocation • Use of a delayed consent strategy
Delayed Consent • Full UK ethical approval • Patients randomised and treated without discussion • Subsequent informed consent in recovery phase • Additional national approval - • Use of data from patients who died before consent
Results - Population 1917 patients scheduled for emergency angiography 29 (1.5%) already randomised in the trial 59 (3.0%) met one or more other exclusion criteria 1829 eligible for recruitment
Results - Population 1917 patients scheduled for emergency angiography 29 (1.5%) already randomised in the trial 59 (3.0%) met one or more other exclusion criteria 1829 eligible for recruitment 1829 Randomised Representative ‘Real-World’ Population
Results - Population Assigned to Heparin 914 915 Assigned to Bivalirudin Received allocated Rx 900 Received no study drug 14 Treatment cross-over 0 LMWH pre-procedure 3 Received allocated Rx 7 Received no study drug Treatment cross-over 4 LMWH pre-procedure
Results - Population Assigned to Heparin 914 915 Assigned to Bivalirudin Received allocated Rx 900 Received no study drug 14 Treatment cross-over 0 LMWH pre-procedure 3 Received allocated Rx 7 Received no study drug Treatment cross-over 4 LMWH pre-procedure Consent not available in surviving patients Consent not available in surviving patients 7 10 Included in analysis 907 905 Included in analysis
Timing of First MACE Event Event curve shows first event experienced
MACE Outcome - Hierarchical Censored by the most significant event - in order displayed
MACE Outcome - Hierarchical Censored by the most significant event - in order displayed
Stent Thrombosis ARC definite or probable stent thrombosis events
Stent Thrombosis ARC definite or probable stent thrombosis events
Primary Safety Outcomes Major Bleed BARC grade 3-5
Safety Outcomes Major Bleed BARC grade 3-5 Minor Bleed BARC grade 2
Study Limitations • Single centre • Potential impact minimised by: • Meticulous trial conduct • Unselected representative population • Study treatments are iv drugs (no ‘skill’ component) • Multiple operators • Outcomes as expected by national norms
Study Limitations • Single centre • Open label • Potential impact minimised by: • Complete follow-up - No ‘lost’ cases • Outcome measures were overt clinical events • Most MI events involved angiographic imaging • Independent blinded adjudication • Open label used in HORIZONS and EUROMAX
Conclusions • A unique study with 100% recruitment of eligible patients
Conclusions • A unique study with 100% recruitment of eligible patients Use of heparin rather than bivalirudin • Reduced rate of major adverse events (NNT = 33) • Fewer stent thromboses and reinfarction events
Conclusions • A unique study with 100% recruitment of eligible patients Use of heparin rather than bivalirudin • Reduced rate of major adverse events (NNT = 33) • Fewer stent thromboses and reinfarction events • Consistent effect across pre-specified subgroups
Conclusions • A unique study with 100% recruitment of eligible patients Use of heparin rather than bivalirudin • Reduced rate of major adverse events (NNT = 33) • Fewer stent thromboses and reinfarction events • Consistent effect across pre-specified subgroups • No increase in bleeding complications
Conclusions • A unique study with 100% recruitment of eligible patients Use of heparin rather than bivalirudin • Reduced rate of major adverse events (NNT = 33) • Fewer stent thromboses and reinfarction events • Consistent effect across pre-specified subgroups • No increase in bleeding complications • Potential for substantial saving in drug costs
Timing of First Major Bleed Event 3 Event curve shows first major bleed experienced
Single-centre Trials ? 6 Common assumptions - based on historic connotations • Smaller studies - often underpowered • Potential subversion of randomisation • Less robust trial procedures and documentation • No adjudication of adverse events
Single-centre Trials ? 7 Common assumptions - based on historic connotations • Smaller studies - often underpowered • Potential subversion of randomisation • Less robust trial procedures and documentation • No adjudication of adverse events No active problems for HEAT PPCI
Single-centre Trials ? 8 Issues related to the patient population • Unselected: External referral to trial centre • Near universal inclusion in trial • Patients typical for UK population • Predominantly Caucasian race
Single-centre Trials ? 8 Issues related to the patient population • Unselected: External referral to trial centre • Near universal inclusion in trial • Patients typical for UK population • Predominantly Caucasian race May affect generalisation to other populations
Single-centre Trials ? 9 Issues related to clinical performance and outcomes In HEAT PPCI - • Randomised treatments are routine iv medications • Established and standardised approach to • Purchase and storage • Administration and dosing • Outcomes are not affected by practice pattern or ‘skill’