Exploring Gaseous and Liquid Compounds: An Overview of Properties and Behaviors
610 likes | 731 Views
This lecture presented by Jasmin Šutković at the International University of Sarajevo discusses the fundamental properties and behaviors of gaseous and liquid compounds. It covers the relationships between pressure, volume, temperature, and the amount of gas as described by the Ideal Gas Law and Boyle's Law. The characteristics of gases and liquids, including their intermolecular forces, unique behaviors, and the nature of phase changes, are explored in detail. The lecture also highlights common gaseous elements and compounds found in various applications.

Exploring Gaseous and Liquid Compounds: An Overview of Properties and Behaviors
E N D
Presentation Transcript
Lecture 7:Gaseous and Liquid compounds Course lecturer : Jasmin Šutković • 17thApril 2014
Contents International University of Sarajevo 1. Gaseous properties and compounds Pressure, Volume, Temperature and Amount relationship The IDEAL GAS Law Mixture of gases Kinetic energy in gases The REAL gas behavior LIQUIDs and its kinetic energy Intermolecular forces in liquids Unique properties of LIQUIDS Vapor pressure CHANGE of states Critical temperatures and pressures
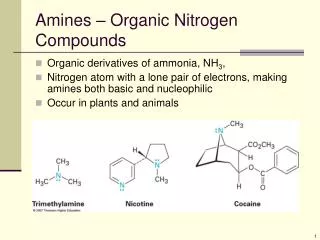
1.Gaseous Elements andCompounds The three common phases (or states) of matter are gas, liquid, and solid • Gases a. Have the lowest density of the three states of matter b. Are highly compressible c. Completely fill any container in which they are placed d. Their intermolecular forces are weak e. Molecules are constantly moving independently of the other molecules present • Liquids a. Dense b. Incompressible c. Flow readily to adapt to the shape of the container d. Sum of the intermolecular forces are between those of gases and solids
Gaseous Elements andCompounds The state of a given substance depends strongly on conditions • Solids a. Dense b. Rigid c. Incompressible d. Intermolecular forces are strong e. Molecules locked in place
PSE location of G,L and S elements The figure of the periodic table shows the locations in the periodic table of those elements that are commonly found in the gaseous, liquid, and solid states
Gaseous Elements andCompounds Many of the elements and compounds are typically found as gases • Gaseous substances include: 1. Many binary hydrides, such as the hydrogen halides 2. Hydrides of the Group-15 elements N, P, and As 3. Hydrides of the Group-14 elements C, Si, and Ge 4. Many of the simple covalent oxides of the nonmetals such as CO, CO2, NO, NO2, SO2, SO3, and ClO2 5. Many low-molecular-mass organic compounds 6. Most of the commonly used refrigerants
Gaseous Elements andCompounds • Gaseoussubstances contain covalent or polar covalent bonds and are nonpolar or slightly polar molecules. • Boiling points of polar compounds are higher than those of nonpolar compounds of similar molecular mass.
Gas Pressure • At the macroscopic level, in order to describe the GAS we requirefour quantities: 1. Temperature (expressed in K or Celsius (C)) 2. Volume (expressed in liters (L) ) 3. Amount (expressed in moles (n)) 4. Pressure (given in atmospheres (atm)) • These variables are not independent — if the values of any three of these quantities are known, the fourth can be calculated.
Units of Pressure Pressure is any force exerted from anything on any surface with which it comes in contact. Units of pressure are derived from the units used to measure force and area For scientific measurements, the S unit for pressure, is the newton per square meter, N/m2, called the paschal (Pa): 1 pascal (Pa) = 1 newton/meter2 (N/m2)
Atmospheric Pressure • Every point on Earth’s surface experiences a net pressure called atmospheric pressure. • Pressure is due to the weight of air above that surface in the atmosphere of Earth (or that of another planet) Air has weight. Imagine a column ( some space) of air that is 2.5 square meter in cross-section that extends vertically above your head until the air runs out in space. At sea level that column of air weighs around 7kg . If you climb mount Everest there is much less air above you in that column, so the weight of air above you will be much less than at sea level.
Athmospheric pressure example This plastic bottle was sealed at approximately 4,300 maltitude, and was crushed by the increase in atmospheric pressure —at 2,700 m and 300 m— as it was brought down towards sea level.
Measurment of A.P • Atmospheric pressure can be measured using a barometer, a closed, inverted tube filled with mercury. • The height of the mercury column is proportional to the atmospheric pressure, which is reported in units of millimeters of mercury (mmHg), also called torr. One (1) bar is atmospheric pressure at sea level. 1atm = 760mmHg = 760torr = 1.01325x10^5 Pa = 101.325 kPa = 14.7 psi
2. The Relationship between Pressureand Volume • Pressure on a gas increases, the volume of the gas decreases ! (gas particles are forced closer together) • Pressure on a gas gas decreases, the gas volume increases ! (gas particles can now move farther apart) • Boyle carried out some experiments that determined the quantitative relationship between the pressure and volume of a gas.
The Relationship between Pressureand Volumen cont.... • Relationship between the two quantities is described by the equation PV = constant (k). • Dividing both sides by P gives an equation that illustrates the inverse relationship between P and V: • This relationship between pressure and volume is known as Boyle’slaw which states that at constant temperature, the volume of a fixed amount of a gas is inversely proportional to its pressure. TEMPERATURE IS CONSTANT
Temperature and Volumen relationship • Hot air rises and gases expand when heated. • Aleksandar Charles carried out experiments to quantify the relationship between the temperature and volume of a gas, he statet his law : At constant pressure, the volume of a given mass of an ideal gas increases or decreases by the same factor as its temperature on the absolute temperature scale. where V is the volume of the gas; and T is the absolute temperature. The law can also be usefully expressed as follows:
The Relationship between Amount of gases and Volumen • Avogadro postulated that, at the same temperature and pressure, equal volumes of gases contain the same number of gaseous particles. • Avogadro’s lawdescribes the relationship between volume and amount of gas: At constant temperature and pressure, the volume of a sample of gas is directly proportional to the number of moles of gas in the sample. • Stated mathematically: Where: V is the volume of the gas(es). n is the amount of substance of the gas. k is a proportionality constant.
In conclusion • The relationships between the volume of a gas and its pressure, temperature, and amount are summarized in the figure below. • The volume increases with increasing temperature or amount but decreases with increasing pressure.
3. Ideal Gas Law • A law relating pressure, temperature, volume, and the amount of an ideal gas. Any set of relationships between a single quantity (suchas V) and several other variables (P, T, n) can be combined into a single expression that describes all the relationships simultaneously. R = 8.3145 J/mol·K (Joule /mol·Kelvin)
Deriving the Ideal Gas Law • A particular set of conditions were chosen to use as a reference; 0ºC (273.15 K) and 1 atm pressure are referred to as standardtemperature and pressure (STP). • The volume of 1 mol of an ideal gas at 0ºC and 1 atm pressure is 22.41L, called the standard molar volumeof an ideal gas.
4. Mixture of gases - Partial Pressures (P) • If the volume, temperature, and number of moles of each gas in a mixture is known, then the pressure exerted by each gas individually, which is its partial pressure, can be calculated. • Partial pressure is the pressure the gas would exert if it were the only one present (at the same temperature and volume). • This law is known as Dalton’s law of partial pressures and can be written mathematically as Pt= P1+ P2+ P3- - - + Pi where Pt is the total pressure and the other terms are the partial pressures of the individual gases.
Partial Pressures cont... • For a mixture of two ideal gases, A and B, the expression for the total pressure can be written as •More generally, for a mixture oficomponents, the total pressure is given by The above equation makes it clear that, at constant temperature and volume, the pressure exerted by a gas depends on only the total number of moles of gas present, whether the gas is a single chemical species or a mixture of gaseous species.
Mole Fractions of Gas Mixtures • The composition of a gas mixture can be described by the mole fractions of the gases present. • Mole fraction ( ) of any component of a mixture is the ratio of the number of moles of that component to the total number of moles of all the species present in the mixture (nt) • Mole fraction is a dimensionless quantity between 0 and 1. • If A = 1, then the sample is pure A, not a mixture. • If A = 0, then sample A is is not pure present in the mixture. The sum of the mole fractions of all the components present must equal 1.
4. Kinetic molecular theory of gases The kinetic molecular theory of gases explains the laws that describe the behavior of gases and it was developed during the nineteenth century by Boltzmann, Clausius, and Maxwell. Kinetic molecular theory of gases provides a molecular explanation for the observations that led to the development of the ideal gas law !!
Several postulates ( rules ) exists but 2 of them are essential for ideal gas behavior : • Postulates 2 and 3 states that all gaseous particles behave identically, regardless of the chemical nature of their component molecules
At ata given temperature, all gases have the same average kinetic energy. • The average kinetic energy of the molecules of a gas is where 2 is the average of the squares of the speeds of the particles and m is the mass of the object. • The square root of 2 is the root mean square (rms)speed (rms) •
Diffusion of Gas •Diffusion is the gradual mixing of gases due to the motion of their component particles even in the absence of mechanical agitation (movement) such as stirring. • Result is a gas mixture with uniform composition - Graham’s law. The ratio of the diffusion rates of two gases is the square root of the inverse ratio of their molar masses. If ris the diffusion rate and M is the molar mass, then r1/r2 = M2/M1 If M1 M2, then gas #1 will diffuse more rapidly than gas #2.
Effusion of Gas •Effusion is the escape of a gas through a small (usually microscopic) opening into an evacuated space. • Rates of effusion of gases are inversely proportional to the square root of their molar masses. • Heavy molecules effuse through a porous material more slowly than light molecules.
The Behavior of Real Gases •Postulates of the kinetic molecular theory of gases ignore both the volume occupied by the molecules of a gas and all interactions between molecules, whether attractive or repulsive. • In reality, all gases have nonzero molecular volumes and the molecules of real gases interact with one another in ways that depend on the structure of the molecules and differ for each gaseous substance.
The Behavior of Real Gases • Real gases behave differently from ideal gases at high pressures and low temperatures. A ) In an ideal gas the interactions between molecules are not accounted B ) in an ideal gas the actual volume taken up by the molecules of gas is not taken into account. So… Molecules of an ideal gas are assumed to have zero volume; volume available to them for motion is the same as the volume of the container.
P,V and T Relationshipsin Real Gases Molecules of a real gas have small but measurable volumes. • At low pressures, gaseous molecules are far apart • As pressure increases, intermolecular distances become smaller • Total volume occupied by gas is greater than the volume predicted by the ideal gas law, so the experimentally measured value of PV/nRTis greater than the value predicted by the ideal gas law.
Liquefaction of Gases Liquefaction of gases is the condensation of gases into a liquid form. • Both the theory and the ideal gas law predict that gases compressed to very high pressures and cooled to very low temperatures should still behave like gases. However, as gases are compressed and cooled, they condense to form liquids.
6. LIQUIDS The Kinetic Molecular Description • Molecules in liquids are very close together, with essentially no empty space between them ( In solids they are more apart) • Molecules in liquids are in constant motion, and their kinetic energy (and hence their speed) depends on their temperature. • Properties of liquids can be explained using a modified version of the kinetic molecular theory.
The Kinetic Molecular Description of Liquids Some characteristic properties of liquids are: 1. Density – Molecules of a liquid are packed relatively close together. – As a result, liquids are much denser than gases – Densities of liquids measured in units of grams per cubic centimeterr(g/cm3) or grams per milliliter (g/mL).
The Kinetic Molecular Description of Liquids cont... 2. Molecular order – Liquids exhibit short-range order because strong intermolecular attractive forces cause the molecules to pack together tightly. – Because of the high kinetic energy of the molecules, they move rapidly with respect to one another. – Arrangement of the molecules in a liquid is not completely random. – Molecules in liquids are ordered because of strong intermolecular attractive forces.
The Kinetic Molecular Description of Liquids cont... 3. Compressibility – Liquids cannot be readily compressed because they have so little empty space between the component molecules. 4. Thermal expansion – Intermolecular forces in liquids are strong enough to keep them from expanding significantly when heated. – Volumes of liquids are somewhat fixed.
The Kinetic Molecular Description of Liquids cont... 5. Fluidity – Liquids can flow, adjusting to the shape of the container, because their molecules are free to move. 6. Diffusion – Molecules in liquids diffuse because they are in constant motion.
Fluidity and Diffusion
Intermolecular forces in liquids • intermolecular interactions, weaker than the intramolecular interactions that hold the atoms together in molecules and polyatomic ions. • Transitions between the solid, liquid, and gaseous states are due to changes in intermolecular interactions but do not affect intramolecular attractions. • Intermolecular forces are electrostatic; they arise from the interaction between positively and negatively charged species.
Intermolecular Forces cont... There are three major types of intermolecular interactions: 1. Dipole-dipole interactions 2. London dispersion forces 3. Hydrogen bonds The first two are described collectively as van der Waalsforces
Intermolecular Forces cont... Dipole-dipole interactions – There are two types of dipole-dipole interactions: 1. Attractive — molecular orientations in which the positive end of one dipole is near the negative end of another (and vice versa) 2. Repulsive — molecular orientations that juxtapose the positive or negative ends of the dipoles on adjacent molecules –The attractive intermolecular interactions are more stable than the repulsive intermolecular interactions
Intermolecular Forces cont... London dispersion forces The London dispersion force is the weakest intermolecular force. TheLondon dispersion force is a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles.
Intermolecular Forces cont... Hydrogen bonds – Molecules that contain hydrogen atoms bonded to electronegative atoms such as O, N, F, and to a lesser extent Cl and S, tend to exhibit strong intermolecular interactions and have high boiling points. – The large difference in electronegativity results in a large partial positive charge on hydrogen and a corresponding large partial negative charge on the O, N, or F atom.
8. Unique Properties of Liquids There are three unique properties of liquids that depend intimately on the nature of intermolecular interactions 1. Surface tension 2. Capillary action 3. Viscosity
Surface Tension • Is the energy required to increase the surface area of a liquid by a specific amount! • Measured as energy per unit area, such as joules per square meter (J/m2) • The stronger the intermolecular forces, the higher the surface tension! • Surfactants are molecules such as soap and detergents that reduce the surface tension of polar liquids like water by disrupting the intermolecular attractions between adjacent molecules
Surface tension example... (a) A paper clip can “float” on water because of surface tension. (b) Surface tension also allows insects such as this water strider to “walk on water.”