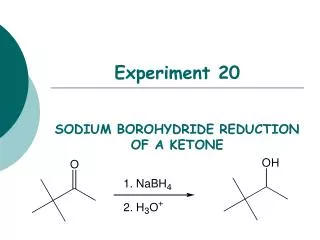
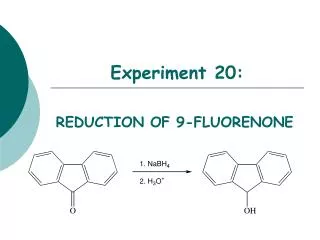
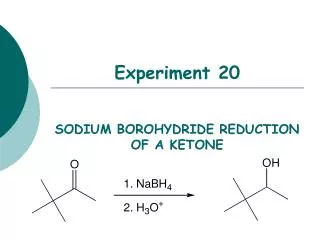
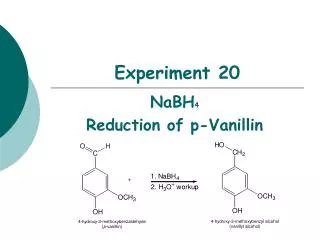
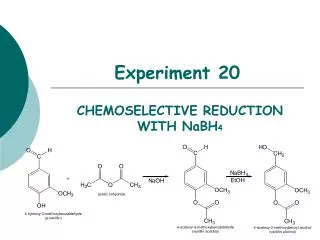
Experiment 20
Experiment 20. Titration of Acids and Bases. In Lab. Work in groups of 4 2 people for Part A; standardization of the prepared solution of NaOH 2 people for Part B; analysis of an unknown acid Each student does their own calculations after sharing data. Part A.

Experiment 20
E N D
Presentation Transcript
Experiment 20 Titration of Acids and Bases CHE118
In Lab • Work in groups of 4 • 2 people for Part A; standardization of the prepared solution of NaOH • 2 people for Part B; analysis of an unknown acid • Each student does their own calculations after sharing data CHE118
Part A • NaOH solution is already prepared, 0.1 M • Do not boil any water • Standardize the NaOH solution to find the next significant figure for molarity 0.1? M (If you use the 0.001 g balance, then you will have 3 SF for the molarity, 0.1?? M) CHE118
Part B • Do not boil any water • There is no weighing bottle; use weighing paper • Use the molarity determined in Part A for calculations in Part B CHE118
Parts A and B • Buret readings have 2 SF after the decimal point 14.18 mL • Obtain volume by difference for titrant (final – initial) CHE118
YouTube VideosHow to Titrate • NAIT Chemical Technology Laboratory Techniques • CarolinaBiologicals • Both are good videos on how to do a titration CHE118
Calculations Part A 1 3 2 g KHP → moles KHP → moles NaOH → M NaOH g → moles; use molecular weight of KHP moles KHP → moles NaOH; use coefficients in balanced chemical equation moles NaOH → Molarity NaOH; M = moles / L 1 2 3 CHE118
Calculations Part B CHE118
Calculations Part B(continued) 4 1 3 2 mL NaOH → moles NaOH → moles KHP → g KHP → % KHP mL NaOH → moles NaOH; M x L = moles, use M from Part A moles NaOH → moles KHP; use coefficients in balanced chemical equation moles KHP → g KHP; use molecular weight of KHP g KHP → % KHP; see previous slide 1 2 3 4 CHE118