Separation t echniques
Separation t echniques. ?. Molecules can be separated : Chemically: b y charge, by action with specific reagents Physically: by solubility, by molecular weight, by density, structure (e.g. shape). Separation. 1. By physical properties. - Centrifugation (centrifugal force).

Separation t echniques
E N D
Presentation Transcript
Molecules can be separated: Chemically:by charge, by action with specific reagents Physically: by solubility,by molecular weight, by density, structure (e.g. shape)
Separation 1. By physical properties - Centrifugation (centrifugal force) • SDS-polyacrylamid-gellectrophoresis (charge) • Agarose-gellectrophoresis (charge) • Gelfiltration (molecular mass) 2. By chemical properties • Ion exchange chromatography • Affinity chromatography • Solubility/desalting • DNA/protein chips or microarrays 3. By physical-chemical properties Native polyacrylamid-gelelectrophoresis Paper-electrophoresis 2D-gelelectrophoresis
Differential centrifugation • - Pellet/supernatant • 2. Density gradient centrifugation • - Continuous gradient • - Step gradient
Gel filtration or size exclusion chromatography Approximately half of the volume of GFC columns is occupied by the resin. Small molecules, which enter all the pores of the resin elute after one column volume. For large molecules, which can not enter any of the pores, at least half of the column volume is necessary. These molecules leave the column after half column volume. Intermediate sized molecules can enter some of the pores, but not all – they elute between the two other fractions. Ball shaped (isometric) molecules are well separated, elongated ones are difficult to separate. Why? Gel filtration does not showgood separation, but can be used for desalting or for the separation of molecules of very different size.
Agarose gel electrophoresis DNA stain agarose Agarose is a polysacharid extracted and purified from sea moss. For separation of large molecules, such as proteins or DNA.
Pulse field gel electrophoresis Continuously changing the direction of the electric field. The large charged particles will not be trapped in the network of polyacrylamide. Very large molecules and particles, even chromosomes can be separated by this technique. The picture shows the separation of chromosomes from different strains of Plasmodium, the parasite causing disease malaria.
Polyacrylamide electrophoresis (PAGE) For separaton of small molecules, such as proteins or small DNA fragments. • Native-PAGE • Molecule mass • Charge SDS-PAGE - Molecule mass + SDS (sodium dodecyl sulphate) + reducing agent (e.g. mercaptoethanol) - No charge difference between molecules - No secondary structure (with reducing agent)
Paper electrophoresis Electrophoresis of proteins can be carried out on wet paper. Though paper electrophoresis does not have a good resolution. However, it is a valuable tool, e.g. it can be used for the separation of native proteins. (in SDS gel electrophoresis the proteins are in denatured state). On this figure we see electrophoretograms of healthy and sick people.
Isoelectric focusing separation on pH-gradient by the charge A - B Forming a pH gradient: Ph gradient 9 8 7 6 5 4 e.g.A With free amfolins or immobilens (weak acidic or basic molecules) Amfolins- forms a pH gradient in electronic field Immobilens-covalently bound to the polyacrylamide matrix e.g.B +
2D-electrophoresis First dimension: Isoelectric focusing Second dimension: Separation by molecule mass Proteins in a breast cancer Proteins in a healthy breast
Ion exchange chromatography of proteins At slightly basic pH (8-9) most proteins are negatively charged and bind to anionic exchangers. When the ion concentration is raised, first those molecules are eluted which have the lowest number of negative side chains. Highly negatively charged proteins can be eluted only with high ionic strength solutes. The figure shows that fractions eluting at different salt concentrations contain very different proteins.
Ion exchange chromatography ANIONS with exchangeable counterions CATIONS with exchangeable counterions starting adsorbtion start desorbtion end desorbtion regeneration buffer counterions separable materials gradient ions
Affinity chromatography Affinity chromatography is a powerful technique to purify one component out of highly complex mixtures. It is based on the interaction of two molecules (biotin-avidin). One is fixed on the column, while the other is selected out of the mixture. A special form of affinity chromatography is metal chelate chromatography, where complexes of heavy metal ions are fixed on the column. These columns have high affinity to proteins or peptides with numerous histidine residues. Elution is carried out by imidazol compounds (His is imidazol-alanine).
Affinity Chromatography immobilization Sample (S) adsorption impurities Desorption of the sample
Affinity separations Affinity chromatography frequently uses antibodies. Another separation technique, which is based on the use of antibodies is magnetic separation. Proteins, organelles, even intact living cells can be separated with colloidal, plastic-coated magnetic beads, carrying antibodies on their surface. The figure illustrates the principle of the separation: mRNA can also separated with Dynabeads coated with oligo(dT) primers. Why?
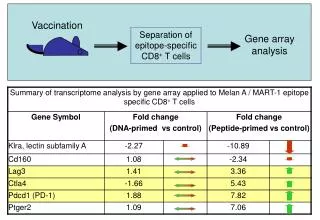
DNA/Protein chips or microarrays For genome wide analysis. DNA chips contains single stranded DNA spots on a glass surface, capable for hybridization with the complementary strand of a cDNA clone reverse transcribed from a single mRNA. One spot represents one gene. There can be 50 000 spots or more on a glass surface. The whole genome can be screened => Genomics In protein chips there are antibodies bound to the glass surface. Each spot recognizes different protein molecules. The whole Proteome can be screened. =>Proteomics