Discovering the Periodic Table: The Language of Chemistry
Explore the Periodic Table and learn about the chemical elements - pure substances that cannot be decomposed by ordinary means - such as Aluminum, Bromine, and Sodium. Understand the arrangement of elements by Dmitri Mendeleev, the groups and periods in the table, and the properties of metals, nonmetals, and metalloids. Discover the characteristics and reactivity of different elements, including alkali metals, alkaline earth metals, transition elements, halogens, and noble gases. Enhance your knowledge of periodic trends, ions, ion charges, and electronegativity with this comprehensive guide to the language of chemistry.


Discovering the Periodic Table: The Language of Chemistry
E N D
Presentation Transcript
The Language of Chemistry • CHEMICAL ELEMENTS - • pure substances that cannot be decomposed by ordinary means to other substances. Aluminum Bromine Sodium
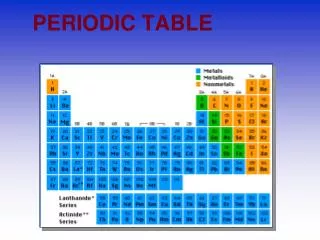
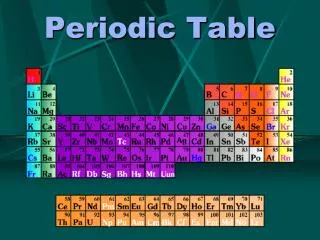
The Language of Chemistry • The elements, their names, and symbols are given on thePERIODIC TABLE
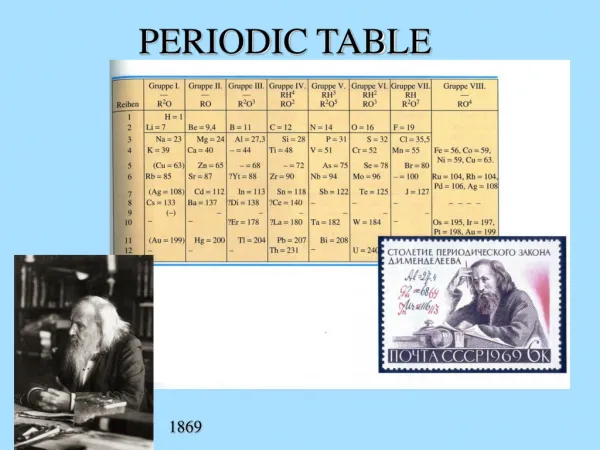
The Periodic Table Dmitri Mendeleev (1834 - 1907) arranged elements in order of increasing atomic mass.
Groups in the Periodic TableVertical columns Elements in groups react in similar ways! Have similar chemical and physical properties
Regions of the Periodic Table 3 classes of elements Metals Nonmetals metalloids
METALS • 80% of elements are metals • Metallic lust, shiny • Good conductors (heat, electricity) • Solid at room temperature (except Hg) • Ductile – can be pulled through wires • Malleable
NonMetals • Most are gases at room temperature • Dull color if solid • P and S are solids • Br - liquid • Poor conductors of heat and temperature • Carbon is the exception
METALLOIDS • Have characteristics of metals and nonmetals • Can be metallic • Brittle (not malleable or ductile • Semiconductors ( Si)
Group 1A: Alkali Metals Silvery colored Very Reactive http://www.youtube.com/watch?v=QSZ-3wScePM Malleable, ductile good conductors soft Cutting sodium metal
Group 2A: Alkaline Earth Metals Magnesium Magnesium oxide Very reactive
Transition Elements Lanthanides and actinides Malleable Good conductors of electricity and heat Iron, Cobalt and Nickel can create magnetic fields Can have several oxidation (Lose Electrons) states
Group 7A: The Halogens (salt makers) F, Cl, Br, I, At Non Metals Extremely reactive/Dangerous Forms salts (NaCl) Forms Acids (HF) Used in: lamps (halogen), Teflon (Flourine + Carbon) http://www.youtube.com/watch?v=u2ogMUDBaf4&feature=related
XeOF4 Group 8A: The Noble (Inert) GasesHe, Ne, Ar, Kr, Xe, Rn • Lighter than air balloons • “Neon” signs • Very Unreactive because they have full electron shells
The Periodic Table -Review 1. Name group 1 2. Name group 7 3. 2 Characteristics of metals 4. 2 characteristics of nonmetals
PERIODIC TRENDS • Atomic size- Increase in atomic number draws electrons closer to the nucleus = smaller
Ionization Energy • The energy required to remove an electron from an atom in order to form an ion • Easy to remove electrons from group 1A more difficult to remove electrons across a row
IONS • IONSare atoms or groups of atoms with a positive or negative charge. • Taking away an electron from an atom gives a CATION with a positive charge • Adding an electron to an atom gives an ANION with a negative charge. • To tell the difference between an atom and an ion, look to see if there is a charge in the superscript! Examples: Na+ Ca+2 I- O-2 Na Ca I O
Forming Cations & Anions A CATION forms when an atom loses one or more electrons. An ANION forms when an atom gains one or more electrons F + e- --> F- Mg --> Mg2+ + 2 e-
PREDICTING ION CHARGES In general • metals (Mg) lose electrons ---> cations • nonmetals (F) gain electrons ---> anions
-3 -2 -1 +1 +2 Charges on Common Ions By losing or gaining e-, atom has same number of e-’s as nearest Group 8A atom.
Ionic Size • Cations are always smaller than the atoms they form • Anions are always larger than the atoms from which they form
Electronegativity • Ability of an atom in an element to attract electrons when the atom is in a compound