Chem Catalyst:
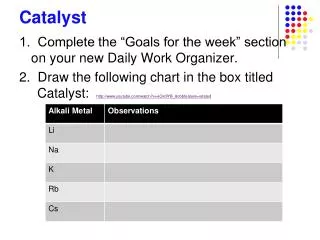
Chem Catalyst:. Quickly draw what you think is inside this balloon. Are they moving in a specific direction?. Homework: None Late work not accepted. Don ’ t ask. TABLE OF CONTENTS: Unit 6: Gas Laws (Write on T of C) 7L – Inquiry Station Lab: Gases 7R – Vocab to Know. Honors Agenda.

Chem Catalyst:
E N D
Presentation Transcript
Chem Catalyst: Quickly draw what you think is inside this balloon. Are they moving in a specific direction? • Homework: • None • Late work not accepted. Don’t ask • TABLE OF CONTENTS: • Unit 6: Gas Laws (Write on T of C) • 7L – Inquiry Station Lab: Gases • 7R – Vocab to Know
Honors Agenda • Vocab Table • Begin, Mini-Lab when done. • Concept Bubble Map using as much vocab words as possible.
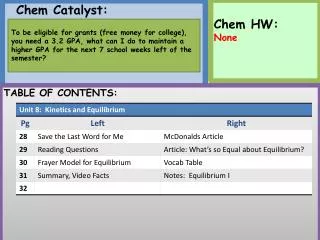
Homework: • Study Guide #1-4, and 20. • Late work not accepted. Don’t ask Chem Catalyst: Using your vocab table, write the difference between heat and temperature? Turn in Test Corrections if done. Parent Letters, Glue Sticks, $1 Notebook. TABLE OF CONTENTS:
Gases • Experience almost no attractive forces. • Easily compressible (can be easily “squished”) 3. Gases have mass.
Gases 4. The distance between gas particles is much greater than the distance between particles of a liquid or solid. 5. Applying pressure to a gas compresses (pushes) particles closer together. 6. Unlike solids and liquids, gases completely fill their containers. 7. Gases have high kinetic energy because their particles move more than particles in a solid or liquid. Kinetic energy is energy due to motion. 8. Gas temperature is measured in Kelvin. Temp measures the average kinetic energy of gas particles in an object.
Kinetic Molecular Theory! • Gas particles are TINY compared to the space between them. So, we do not count the volume of the particles. Itty-bitty Space between = BIG Itty-bity
KMT continued • The particles are in constant RANDOM motion. The particles collide with the walls of the container. These collisions result in pressure. Exerts pressure
More KMT! • The gas particles do not attract or repel each other. They move randomly. • The average kinetic energy of the gas is proportional to the Kelvin temperature. Kinetic Energy Temperature
Honors: Why use Kelvin? • Gases exist at temperatures lower than 0°C. It doesn’t make sense for gases to move at negative temperatures. • So Kelvin was created where 0K is absolute zero, the lowest temperature that can exist, where molecular motion stops.
K = °C + 273 • Convert room temperature (20°C) to Kelvin. • Convert standard temperature (0°C) to Kelvin. • Convert 254 K to Celsius. • Convert 40°C to Kelvin. • Convert 300 K to Celsius.
Pressure • Caused by gases colliding with container. • Units include • 1 atmosphere = 1 Torr = 760 mmHg = 101kPa
Volume • Usually in liters. • Honors, Memorize: 1L = 1 dm3
Standard Temp and Pressure • Reference to common conditions of a gas at 0°C (273K) and 1 atm.
Honors: Effusion and Diffusion • Effusion: rate at which a gas escapes through a small opening in a container • Diffusion: rate at which a gas travels across a room/container from high to low concentration.
Gas Laws • Shows relationships between Pressure, Temperature, Volume, and moles of gas particles. • There are FOUR laws to understand. Formulas will be given to you.
Boyle’s Law • Has NOTHING to do with temperature! • Shows the relationship b/w pressure and volume of a gas when moles and temperature are constant. • Demo 1: Sit on Balloon • Demo 2: Marshmallow in Syringe • Pressure Volume (inversely proportional) • Formula: P1V1 = P2V2 1= before 2= after
Graph of Boyle’s Law V and P are inversely proportional! As one goes up the other goes down!
Example 1 • A balloon has an initial volume of 1L and pressure at 1 atm. When you sat on the balloon, its volume decreased to 0.5L. What is the pressure inside the balloon after you sat on it? • By decreasing volume, we should get a higher or lower pressure according to Boyle’s Law? Does the answer make sense?
Practice Problems • A gas has a volume 2.0 L and a pressure of 1.0 atm, what volume will the gas have at 4.0 atm? At constant temperature. • A gas has a volume 3 L and a pressure of 2.0 atm, what pressure will the gas have at 24 L? At constant temperature. • A gas has a volume 50.0 L and a pressure of 8.0 atm, what volume will the gas have at 10.0 atm? • A gas has a volume 20.0 L and a pressure of 5.0 atm, what pressure in mmHg will the gas have at 25.0 L?
Homework: • Bring a few uncrushed cans. • On 10L, Summarize Boyle’s Law in 4 sentences. Chem Catalyst: 1) What is absolute zero? 2) What happens to the kinetic energy of gas particles when you decrease the heat? 3)Would pressure go down or up? Glue Sticks, $1 Notebook. TABLE OF CONTENTS:
Diffusion: rate at which a gas travels across a room/container from high to low concentration.
Agenda • Correct HW (10 min) • BL #1-5 on your own (15 min) • Go over it (5 min) • Dry Erase Races, Males VS Females!!!!
Exit Ticket (1/2 sheet of paper) Turn this in • What is the coldest possible temperature? What happens at this point? • What are the temperature and pressure for STP? • What does Boyle’s Law say about the relationship between pressure and volume? • A sample of gas in a syringe has a volume of 10 L and a pressure of 3 atm. What is the new volume if the pressure is decreased to 1.5 atm?
Homework: • Study Guide Q’s 12-14, 19, 26 Chem Catalyst: Explain why people tend to put air into their tires during the winter and not during the summer? Glue Sticks, $1 Notebook. TABLE OF CONTENTS:
Honors Relay Race Questions • What is absolute zero value? • At absolute zero, what happens at the molecular level? • It is 5 K outside. What is the temperature in oC? • It is 20oC, what is the temperature in Kelvin? • What does Boyle’s Law state? • If I increase the volume will pressure increase or decrease? • I start with a gas that is 5 atm and a volume of 500 mL. If the final volume is 1500 mL, then what is the final pressure be in mmHg. • A 20.0 L tank of nitrogen has a pressure of 2.00 atm. Calculate the volume in dm3 of nitrogen if its pressure is changed to 12.0 atm while its temperature remains constant. • What is standard temperature and pressure values?
Relay Race Questions • What is absolute zero? • It is 5 K outside. What is the temperature in oC? • It is 20oC, what is the temperature in Kelvin? • What does Boyle’s Law state? • If I increase the volume will pressure increase or decrease? • I start with a gas that is 5 atm and a volume of 500 mL. If the final volume is 1500 mL, then what is the final pressure? If in Honors, I want the pressure in mmHg. • Honors: A 20.0 L tank of nitrogen has a pressure of 2.00 atm. Calculate the volume in dm3 of nitrogen if its pressure is changed to 12.0 atm while its temperature remains constant. • What is standard temperature and pressure?
Quickwrite #2: What do you notice? Bullet points are fine. Draw Graph and explain it. Directly/indirectly proportional? Constant:
Charles’s Law • Shows the relationship between volume and temperature where pressure and moles of gas remain constant. • Demo: Balloon under ice water? Shrinks • Temperature Volume (directly proportional) • Formula: T MUST be in Kelvin!!!!
This is an direct relationship! As one goes up the other goes up!
Example 1: A can had an initial volume of 5L and temperature of 250oC. The temperature of the can then cooled to room temperature (20oC), what will the final volume of the can be? Why does temperature increase volume at the molecular level?
You Try and Discuss with Partner(s) A 2.5L balloon at T = 25oC increases in volume at 45oC, what is the new volume of the balloon? According to Charles’s Law, increasing temp increases volume, does the answer justify this law?
Next to your home group, work together on the following: • Study Guide Questions #9 10, 11, 16, 17, and 18 • Group Leaders, make sure EVERYONE understands. • When done, check your answers with Mr. Narez
Procedures: • Add 30mL of water to the inside of the can. • Place flame under can using tongues until water VIGOROUSLY boils, steam must come out • Carefully turn the can face down into the cold water bath. On 11L, Write, “Can Crush Demo” Write a 3-4 sentence hypothesis about what you think happened to the soda. What is happening at the molecular level? What observations would help support your hypothesis?
Can Crush Explanation Below your first paragraph, paraphrase the following: • Before heating, can had air and water inside. • By boiling, kinetic energy increases and water turns into vapor causing the air to leave the can. • When submerged in water, the vapor turned back into water which does not take up the shape of the container so there’s all this empty space. • The air pressure outside the can is greater causing the can to compress.