Atomic Structure
This comprehensive guide explores atomic structure, focusing on isotopes and their properties. It defines protons, neutrons, electrons, and delves into concepts like atomic number (Z), mass number (A), and relative atomic mass (Ar). The relationship between isotopes and chemical properties is highlighted. Additionally, the guide covers ionization energy, balanced chemical equations, and the formation of salts through acid-base reactions. Ideal for students and chemistry enthusiasts alike, this resource clarifies fundamental concepts crucial for mastering atomic theory and chemical interactions.

Atomic Structure
E N D
Presentation Transcript
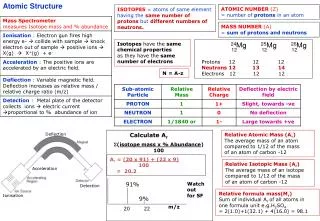
Atomic Structure 26Mg 25Mg 24Mg 12 12 12 Protons 12 12 12 Neutrons 12 13 14 Electrons 12 12 12 ATOMIC NUMBER (Z) = number of protons in an atom ISOTOPES = atoms of same element having the same number of protons but different numbers of neutrons. MASS NUMBER (A) = sum of protons and neutrons Isotopes have the same chemical properties as they have the same number of electrons N = A-z Deflection Calculate Ar Relative Atomic Mass (Ar) The average mass of an atom compared to 1/12 of the mass of an atom of carbon -12 (isotope mass x % Abundance) 100 Ar = (20 x 91) + (22 x 9) 100 = 20.2 Relative Isotopic Mass (Ar) The average mass of an isotope compared to 1/12 of the mass of an atom of carbon -12 Acceleration 91% Watch out for SF Detection Relative formula mass(Mr) Sum of individual Ar of all atoms in one formula unit e.g.H2SO4 = 2(1.0)+1(32.1) + 4(16.0) = 98.1 Ionisation 9% m/z 20 22
Empirical Formula n = mol M = Mass Mr= molar mass aAbB NaOH + Cl2 NaCl + NaClO + H2O Na= 1 2 x2 on Left 2 NaOH + Cl2 NaCl + NaClO + H2O Cl= 2 2 balanced 2 NaOH + Cl2 NaCl + NaClO + H2O H = 2 2 balanced 2 NaOH + Cl2 NaCl + NaClO + H2O V O = 2 2 balanced n 24 2 NaOH + Cl2 NaCl + NaClO + H2O n C V p n NA EF = simplest whole number ratio of atoms in one molecule MF = Actual number of each type of atom in one molecule m Use a moles triangle to 1 Mr n Calculate moles of A = nA Use Balanced Equation Mole ratio to find moles of B A compound 34.33% Na, 17.91% C, 47.76% O, has a Mr =134 -Find EF and MF n = mol C = concentration V = Volume (dm3) 2 b x nA = nB a Na C O 34.33 17.91 47.76 34.3317.9147.76 23.0 12.0 16.0 =1.493 = 1.493 = 2.985 1.4931.4932.985 1.493 1.493 1.493 = 1 =1 = 2 EF = NaCO2 EF mass = 23+12+32 = 67 % Use a moles triangle by Ar by smallest Calculate required quantity of B V, C, m, p etc.. 3 n = mol V = gas Volume (dm3) 24= Volume of 1 mol (dm3) V is in dm3 dm3 to cm3 x by 1000 cm3 to dm3 by 1000 n = mol p = particles NA =6.022 x1023 EF x N = MF N = Mr EF mass N = 134 = 2 67 Moles & Equations MF = Na2C2O4 Balanced Equations H2SO4 -Sulfuric acid, NH3 -Ammonia HNO3 -Nitric acid NaOH –sodium hydroxide Non metal elements Are X2 e.g. O2H2 Cl2, N2 1-Balance METAL 2-Balance NON metal 3-Balance H and O last which ever of O and H is in fewest substances first)
IE Ionisation Energy: The energy needed to remove 1 mole of electrons from 1 mole of gaseous atoms forming 1 mole of gaseous positive ions. Successive Ionisation Energies for Ca First shell 1s2 Closest to nucleus Most difficult to remove 2 Second shell 2s2 2p6 First IE = M (g) M+(g) + e- Second IE = M+(g) M2+(g) + e- IE1 8 Decreases down group Third shell 3s2 3p6 Group 2 xs2 full s sub-shell 8 Group 3 xs2 xp1 full s sub-shell shields outer p1 further from Nucleus “big jump” in IE Shows when a new Inner shell is started Increases across period 2 Group 5 xs2 xp3 half Full p sub-shell no spin pairing successive removal of e- Outer shell 4s2 Furthest from nucleus Most shielding Easiest to remove Each successive IE is greater as there are more protons to hold fewer electrons. 0 1 2 3 4 5 6 7 0 1 Group 6 xs2 xp4 spin pairing causes repulsion between electrons –lowers IE Group Number Increases across period: Increased nuclear attraction Electron shielding –says the same Atomic radius decreases Decreases down group: Increased nuclear attraction outweighed by electron shielding Atomic radius increases Electron Structure Energy/distance from Nucleus Big jump after 5th IE shows where electron Is removed from a full inner shell,so elements in group 5) Electrons only pair up If they can not occupy orbital singly 4p Electron Pair with Opposite spin Each orbital can hold two e- 3d 4s 4s filled before 3d 4s emptied before 3d s p 3p 3s 2p s p 26Fe atom 26 electrons 2s d = 1s22s22p63s23p64s23d6 Order of Filling Loss of outer shell 4s2 first 1s22s22p63s23p64s23d104p6 Fe ions Highest energy Occupied orbital Identifies the block 1s Fe2+ ion = 1s22s22p63s23p6 3d6 Fe3+ ion = 1s22s22p63s23p63d5 Other electrons then lost from inner 3d
Acids, Bases, and Salts Acid + Carbonate Salt + water + CO2 2HCl + MgCO3 MgCl2 + H2O + CO2 Effervescence Solid dissolves Acid = Proton Donor Base = Proton Acceptor Acid + MetalSalt + Hydrogen HCl + Mg MgCl2 + H2 (g) Effervescence Metal dissolves Redox Acids H2SO4 = Sulfuric acid HCl = Hydrochloric acid HNO3 = Nitric acid a Salt e.g. MgCl2 Acid + Base Salt + water HCl + MgO MgCl2+ H2O Salt =When the H+ of an acid is replaced by a metal or ammonium ion NH4+ Acid + Alkali Salt + water 2HCl + Mg(OH)2 MgCl2 + 2H2O Bases NaOH = sodium hydroxide KOH = potassium hydroxide NH3 = Ammonia e.g. H2SO4 + 2NH3 (NH4)2SO4 e.g 2HNO3 +Ca(OH)2 Ca(NO3)2+ 2H2O A salt consists of a Positively charged Cation–from a metal, or Ammonium ion And a Negatively charged Anion –which is the remains of the acid When 4.76g of hydrated cobalt chloride were heated, they gave 2.60g of anhydrous cobalt chloride (CoCl2). Cation Salt Anion Acid Mg Mg SO4 H2 SO4 Mg2+ + SO42- Magnesium sulfate Sulfuric acid Magnesium ion Sulfate (VI) ion NH3 1) Mass of water lost = 4.76-2.60 = 2.16 g NH4 NO3 HNO3 NH4 + + NO3 - CoCl2 .XH2O (s) CoCl2 (s) + X H2O (g) Ammonium nitrate Nitric acid Ammonium ion nitrate(V) ion 4.76 g 2.60g 2.16 g An anhydroussalt MSO4 Has lost its water of Crystallisation 2) Moles of substances (= m/Mr) CuSO4.5H2O (s) CuSO4 (s) + 5H2O (g) 2.60 129.9 2.16 18 Hydrated salt –blue crystals Anhydrous salt -white powder = 0.02 = 0.12 A Hydrated salt MSO4.X H2O Has water of crystallisation 3) Divide by smallest gives whole number ratio X H2O = Water of Crystallisation A number of Water molecules bonded in the crystal Shown as a dot formula CuSO4 .5H2O MgSO4 .7H2O Na2CO3. 10 H2O = 0.02 = 0.12 0.02 0.02 = 1 = 6 CuSH10O9gives a dot formula of CuSO4. 5H2O CoCl2 .6H2O
0 +1-2 +1-2+1 0 O.N. change Na + H2O NaOH + H2 Oxidation + 0 - Na: 0 to +1 = Oxidised H : +1 to 0 = Reduced 0 +1-2 +1-1 +1+1-2 increases Cl2 + H2O HCl + HClO Cl 0 to +1 = Oxidation Cl 0 to –1 = Reduction decreases Disproportionation = Oxidation and reduction of the same species Reduction OIL RIG Oxidation = Loss of electrons Reduction = Gain of electrons Redox Oxidation number(state) The number of electrons that have to be gained or lost for an atom to acquire noble gas configuration. e.g. Cl =(Ne) 3s23p5 has to: gain 1 (-1) or lose 7 (+7) Other states are possible in between the extremes • Oxidation Number of S in H2SO4 • element, Ion,or Compound =compound so Sum of O.N = 0 • H+H+S+O+O+O+O =0 • Using rules +1+1+S-2-2-2-2 = 0 • As a sum +2+S-8 = 0 • S = +6 Oxidising Agents gain electrons and get Reduced e.g. Cl2 , H2SO4 O2 MnO4-, HNO3 Reducing Agents lose electrons and get oxidised e.g. Na, H2, Fe2+ CO, C, 0 +1 –1 +2-1 0 Mg + 2HCl MgCl2 + H2 Mg: 0 to +2 = Oxidation H : +1 to 0 = Reduction Oxidation number in names shown as a Roman Numeral Iron(III) = Fe+3, Nitrate(V) = NO3-, Chlorate(I) =ClO- sulphur(IV) oxide = SO2
Ionic Intermolecular Forces Covalent bonds only : : H – O: H – O: H H + - . . . . . . . x x x x x x x + . x H . . . x x H N . 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ H Group 1 or 2 Dot and cross Diagrams Show Outer electron and charges brackets Electrostatic attraction between oppositely charged ions Hydrogen Bonds Van-der Waals forces + - - + : 2+ 2- Conduct electricity Whenmelted or Dissolved as ions free to move Strongest IMF A permanent dipole force found in compounds that have F-H, O-H, or N-H bonds Mg O Ions form by transfer of electrons from metal to Non metal : e.g. NaCl, MgO • Uneven electron distribution • causes a temporary dipole. • This induces a dipole on • adjacent molecule • Temporary dipoles attract + - Covalent A Shared pair of electrons Dot and cross diagrams show outer electrons only : H-bond .. .. .. Lone Pair : 2 molecules Lone Pairs Dipoles Labelled bond • The only IMF in non- polar • molecules e.g. I2, Cl2, F2, CH4 • In compounds with • C-C, C-H, Cl-Cl, F-F, I-I bonds • stronger in molecules with more • electrons O C O H O Between non-metals electrons contribute to outer shell of both atoms x .. .. + . . Bond Pair x Dative Covalent (coordinate) H Attraction between + H of one molecule and a lone pair from a neighbouring electronegative atom, O, or N or F + Molecules Are made from Atoms covalently Bonded together Ammonium ion NH3 +H+ NH4+ Permanent Dipole Forces H Covalent bonds have permanent dipoles due to differences in Electronegativity Both bond electrons from one atom (N) Bp Bonding Actual Bp x x H2O C-Cl, C-N, C-O,H-Cl, H-Br, C=O Metallic H2Te + - + - x H2Se Bond strength increases As electron density increases H – Cl H – Cl x Positive ions attracted to sea of delocalised electrons H2S x The attraction between the + dipole of one bond to the - dipole of another Expected Bp Hydrides Of Group 6 Bond strength decreases As electron density decreases Anomalous properties of water due to H-Bonding O=C=O In Symmetrical moleculesdipoles cancel so overall molecule is Non-polar e.g. CO2, BF3, CCl4 Conduct electricity due to mobile delocalised electrons 1) Water has a higher Bp than expected 2) Ice is less dense than water H-bonds are longer so water molecules further apart - High Mp/Bp Strong bonds Malleable and Ductile O In unsymmetricalmolecules dipoles do not cancel e.g. H2O, NCl3, CHCl3 + + H H
Shapes of Molecules & Ions N H 104.5o N xx xx H • Shape is determined by the number of electron pairs • or groups around the centre atom of the molecule • Electron pairs repel each other as far away as possible • to minimise repulsion between them. In multiple bonds the shape is determined by the number of groups of electrons that are bonded to the Centre atom CO2 = 2 –Linear NO3- = 3 –Trigonal plane MnO4- = 4 -Tetrahedral Effect of Lone Pairs: Lone Pairs repel Bond Pairs more strongly and so bond angles are decreased by 2.5o for each Lone Pair Delete two bonds and replace with Lone Pairs In ions with a positive charge e.g. PCl4+ , NH4+ subtract one Electron from the total before calculating number of Electron Pairs
Uses of Group 2 compounds Thermal decomposition of carbonate Mg Mg Group Properties (Ne) 3s2 Atomic Radius Melting Point Heat Medical: Mg(OH)2 “milk of Magnesia” Ant acid indigestion remedy -neutralises stomach acidity MCO3 MO + CO2 Ca Ca Stability of carbonate increases down group Reactivity (Ar) 4s2 Ionisation energy Sr Group 2 Oxide + Acid CaSO4 –Gypsum, “Plaster of Paris” Sr MO + 2 HNO3 M(NO3)2 + H2O (Kr) 5s2 Ba BaSO4 “Barium meal” Before X-Ray Observation: Solid Dissolves –No effervescence Metallic bonds are weaker as sea of delocalised electrons more diffuse so less energy needed to melt metal Increased nuclear attraction outweighed by more electron shielding. Outer electrons are further away and easier to loose Ba (Xe) 6s2 Acidbase reaction forms a SALT MO is a base (proton acceptor) HNO3 is a proton donor (acid) Agricultural: Ca(OH)2 “slaked Lime” Use: Neutralise soil acidity Excessive use makes soil Too alkaline Group 2 • s-block • metals • Have s2 outer • shell electrons • All form M2+ • ions • Oxidation state • +2 • in compounds • Alkaline earth • metals Lime Chemistry Metal + acid Metal + H2O Limestone M + 2 HCl MCl2 + H2 (g) CaCO3 Heat M + H2O M(OH)2 + H2 Lime CaCO3 CaO + CO2 Observations CaO Observations H2O • Effervescenceof H2(g) • Metal dissolves • Effervescence of H2(g) • Metal dissolves Slaked lime CaO + H2O Ca(OH)2 Ca(OH)2 Mg only reacts with steam 2Mg + 2H2O 2MgO + H2 Metal + O2 CO2 Metal + O2 “milky” Test for CO2 Ca(OH)2 + CO2 CaCO3 + H2O 2 M + O2 2 MO Burns with Bright light CaCO3 M(OH)2more soluble Down the group pH increases as concentration of OH- increases XS CO2 Oxide is soluble in water (more down group) CaCO3 + CO2 +H2O Ca(HCO3)2 Ca(HCO3)2 MO + H2O M(OH)2 pH of solution =12 Calcium hydrogencarbonate
Reactions and Uses of Cl2 Physical Properties Trends -1 +1 0 Cl2 + H2O HCl + HClO Cl2 used in drinking water– it kills germsbut is toxic F2 (g) Electronegativity Mp /Bp Disproportionation Cl2 Oxidising power Atomic Radius Cl is oxidised from 0 to +1 and reduced from 0 to -1 (g) Reactivity Cl2 reacts with cold dilute NaOH Disproportionation Br2 0 -1 +1 (l) Cl2 + 2 NaOH NaCl + NaClO + H2O Decreases Down Group Increases Down Group I2 NaClO= Sodium chlorate(I) =Bleach (s) Cl is oxidised from 0 to +1 and reduced from 0 to -1 This solution will turn blue litmusred (HCl) then bleaches white (NaClO) Bigger atoms with more electrons have more electron shielding. stronger Van der Waals forces so more energy needed to break bonds Smallest atom with least electron shielding and most effective nuclear charge attracts electrons most strongly Cl2 reacts with hot concentrated NaOH Disproportionation 0 -1 +5 Cl2 + 6 NaOH 5NaCl + NaClO3 + 3H2O Group 7 Cl is oxidised from 0 to +5 and reduced from 0 to -1 Displacement Reactions Tests for Halide ions with Ag+ A higher halogen (more oxidising) will oxidise a lower halide ion. The Oxidising Halogen gets reduced. red 0 -1 Dilute nitric acid added to stop other silver compounds from precipitating Silver Nitrate (AgNO3)solution is added X2 + 2Y- 2X- + Y2 -1 0 ox Ionic equation observation Non-polar solvent e.g. hexane Solubility In ammonia Ionic equation Cl2 + 2Br- 2Cl- + Br2 Ag+ + F- AgF (aq) F- Colourless solution becomes yellow solution Colourless solution orange (s) Soluble in dilute NH3 Cl- Ag+ + Cl- AgCl (s) Cl2 + 2I- 2Cl- + I2 White ppte Colourless solution becomes Brown solution (s) Soluble in concentrated NH3 Br- purple Ag+ + Br- AgBr (s) Cream ppte Br2 + 2I- 2Br- + I2 Colourless solution becomes Brown solution insoluble in concentrated NH3 (s) I- Ag+ + I- AgI (s) purple Yellow ppte
Periodicity • Patterns of physical & chemical behaviour repeat from period to period • Physical properties show gradual changes depending on the structure and bonding, and particularly • NP = Number of Protons (Nuclear attraction) • ES = Electron Shielding due to full inner electron shells. • AR = Atomic Radius or size of the atom- proximity of nucleus to outer shell. Atomic Radius First Ionisation Energy Electronegativity 0.25 3.5 1600 3 1400 Melting Point 0.2 -1 1200 2.5 / kJ mole 1000 0.15 1800 2 Atomic Radius / nm 800 Electronegativity Index 1600 I 1 1.5 0.1 1400 600 1 1200 400 0.05 1000 0.5 200 Melting Point / K 800 0 0 0 600 10 11 12 13 14 15 16 17 18 10 11 12 13 14 15 16 17 18 10 11 12 13 14 15 16 17 18 400 Atomic Number (Z) Atomic Number (Z) Atomic Number (Z) 200 Trend: Increases NP: Increases ES: Constant AR: Decreases Bonding electrons are attracted more and are closer 0 10 11 12 13 14 15 16 17 18 Atomic Number (Z) Atomic Radius 0.25 Electronegativity M Pt /K Melting Points 0.2 2000 1.6 1.4 Atomic Radius /nm 0.15 1500 1.2 Electronegativity Index 1.0 0.8 1000 0.1 0.6 0.4 500 0.05 0.2 0 0.0 0 Be Mg Ca Sr Ba Be Mg Sr Ba Ca Be Mg Ca Sr Ba Across Period Number of Protons Increasing Electron Shielding constant Conductivity 1.2 1 0.8 0.6 Conductivity relative to Al 0.4 0.2 0 10 11 12 13 14 15 16 17 18 Atomic Number (Z) Trend: Decreases NP: Increases ES: Constant Electrons attracted more strongly so atoms decrease in size Trend: Increases NP: Increases ES: Constant AR: Decreases Electrons attracted more strongly so more energy needed to remove outer e- Trend: Increase- decrease Giant lattice structures Na-Al – Strong metallic bonds Si –vStrong Covalent bonds Simple Molecular structures P –Ar -weak Van der Waals forces easily broken Trend: Inc -Dec-None Na-Alnumber of delocalised electrons per atom increases Si –semi-conductor P –Ar – None as no Delocalised electrons Down Group Electron Shielding Increasing outweighs Increasing Number of protons Chemical properties are similar for Group elements as they have a similar outer shell electron arrangement First Ionisation Energy 1000 800 I.E.1 /kJ mole-1 600 400 200 0 Be Mg Ca Sr Ba Trend: Increases NP: Increases ES: Increases ES outweighs NP More full shells, Electrons further away attracted less strongly Trend: Decreases NP: Increases ES: Increases AR: Increases Electrons further away attracted less strongly so less energy needed - Trend: Decreases NP: Increases ES: Increases AR: Increases Bonding electrons further away attracted less strongly Trend: Decrease AR: Increases Metallic bonding is weaker electron density in delocalised sea decreases so Less attraction for positive ions