WATER
WATER. Water Water Everywhere But Nary a Drop to Drink ( Coleridge.Rime of the Ancient Mariner). Earth’s surface – 70% water 97% of water in oceans: salt water 3% is fresh water 25% of fresh water is groundwater 75% of fresh water is in ice and glaciers

WATER
E N D
Presentation Transcript
Water Water Everywhere But Nary a Drop to Drink (Coleridge.Rime of the Ancient Mariner) • Earth’s surface – 70% water • 97% of water in oceans: salt water • 3% is fresh water • 25% of fresh water is groundwater • 75% of fresh water is in ice and glaciers • Only .03% of water is available to us: in atmosphere, streams, rivers, wetlands, lakes • (Some is lost to pollution)
DESALINATION • DISTILLATION • Water boiled, steam is condensed, salt left behind, (brine liquid). • Requires much energy REVERSE OSMOSIS - Water forced through impermeable membrane - Salt left behind (brine liquid) CONSEQUENCES - Salt deposits on land harm plants and animals - Dumped in bay or coastal areas harms fish and aquatic life
WATER USAGE • How do we consume water? • Agriculture 70%, Industry 20%, households 10% • Why do we put drinking water in our toilets? • What is grey water and contaminated water? • Gray water: showers, washing machines, bathroom sinks – can be used for watering plants, washing cars, flushing toilets • Contaminated water – from toilet, kitchen sinks, dishwashers
PROPERTIES OF WATER • Liquid, solid, and gas—at the temperatures found on Earth. • Universal Solvent – dissolves more substances than other liquid: carries valuable chemicals, minerals and nutrients through ground and our bodies. • pH is neutral 7 (neither acidic or alkaline)
Water Density • When cooled from room temperature, liquid water becomes increasingly dense (as with other substances.) • However, at 4C or 39F pure water reaches its maximum density and then starts to expand and becomes less dense • This is why ice floats. If not the bottom of our lakes would freeze. • High water density allows sound to move through it over long distances (important for whales).
PROPERTIES OF WATER • HIGH SPECIFIC HEAT INDEX • – absorbs a lot of heat before it gets hot. • Makes a good coolant in car radiators and for industry • Regulates rate at which air changes temperature so temperature change between seasons is gradual. AIR PRESSURE AFFECTS Boiling point at sea level is 212 F; at 5000 ft. 202.9 F (With less atmospheric pressure it takes longer to boil an egg) CONDUCTIVITY Pure water does not conduct electricity until it has dissolved the inorganic substances around it. (positively or negatively charged ions) A failing sewage system would raise the conductivity because of the presence of chloride, phosphate, and nitrate; an oil spill would lower the conductivity.
Conductivity of Water Pure water does not conduct electricity until it has dissolved the inorganic substances around it. E.g. chloride, nitrate, sulfate, and phosphate anions (ions that carry a negative charge) or sodium, magnesium, calcium, iron, and aluminum cations (ions that carry a positive charge). Organic compounds like oil, phenol, alcohol, and sugar do not conduct electrical current very well and therefore have a low conductivity when in water. A failing sewage system would raise the conductivity because of the presence of chloride, phosphate, and nitrate; an oil spill would lower the conductivity.
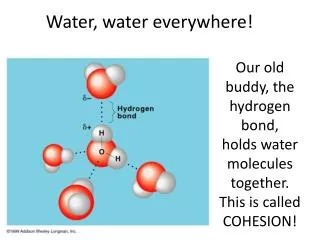
SURFACE TENSION • Water has a very highsurface tension, a property of the surface of a liquid that allows it to resist an external force. • COHESION: water molecules attracted to other water molecules creates the thin “skin” along the surface of water Surface tension: • Allows water striders to “walk on water” ADHESION: water molecules attracted to other materials. - Allows the capillary action that carries water and its dissolved substances to move through the roots of plants and through the tiny blood vessels in our bodies.
FOAM IN LAKES AND STREAMS • Foam accumulates against logs, banks of streams, lake shores. Starts out white, turns brown • Surfactants (either anthropogenic: e.g.detergents, shampoos, or naturally produced from algae and plants) are molecules that are called surface active agents that interact with water reducing the surface tension • Foam is produced as air, introduced in the turbulence of stream riffles .... bubbles to the water surface where surface tension has been reduced.