Carboxylic Acids
Carboxylic Acids. Unit 6. General Formula. RCOOH and ArCOOH Functional Group = O C -OH. Common Names…. 1C – Formic Acid ( Methanoic Acid) From Formaldehyde ( methanal ) 2C – Acetic Acid ( Ethanoic Acid) From Acetaldehyde ( ethanal ) 3C – Propanoic Acid ( Propanoic Acid)

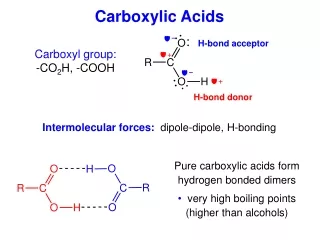
Carboxylic Acids
E N D
Presentation Transcript
Carboxylic Acids Unit 6
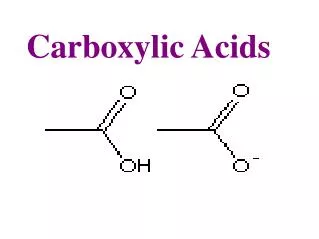
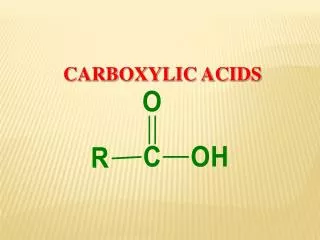
General Formula RCOOH and ArCOOH Functional Group = O C -OH
Common Names… 1C – Formic Acid (Methanoic Acid) • From Formaldehyde (methanal) 2C – Acetic Acid (Ethanoic Acid) • From Acetaldehyde (ethanal) 3C – Propanoic Acid (Propanoic Acid) • From Propanaldehyde (propanal) 4C – Butyric Acid (Butanoic Acid) • From Butyraldehyde (butanal)
Uses – Just FUN Facts – Not Tested! • Formic Acid – First Isolated from Ants • Used in dyeing and finishing textiles and many other uses! • Acetic Acid – Naturally produced by us • Aka vinegar – many uses • Propanoic Acid – in Greek, means “first fat” • Grain Preservative and antifungal • Butyric Acid – Naturally occurring – Greek for butter • Smells like vomit
Preparation of Carboxylic Acids – You already know this • Oxidation of aldehydes O KMnO4 O R-C-H R-C-OH RCHO KMnO4 RCO2H
Example • Form Pentanoic Acid (S,C,N)
Reactions of Carboxylic Acids • Halogenation: RCO2H + X2 RXCO2H + HX ArCO2H + X2FeX3 ArXCO2H + HX Form 2,3,4 – trichloroheptanoic acid (str,cond) O Cl Cl Cl O C-C-C-C-C-C-C-OH + 3Cl2 C-C-C-C-C-C-C-OH +3HCl CH3(CH2)5CO2H + 3Cl2CH3(CH2)2(CHCl)3CO2H + 3HCl
Examples of Halogenation Form paraiodobenzenoic acid (str) + I2FeI3 + HI I
Reactions of Carboxylic Acids 2. Nitration of benzanoic acid ArCO2 + HNO3H2SO4 ArNO2CO2H + H2O Form metanitrobenzanoic acid (str,cond)
Reactions of Carboxylic Acids Acid Salt formation can be done 2 ways… • RCO2H + M° ArNO2CO2H + H2O Metal can be same 7 as before
Examples React ethanoic acid with sodium (str,name,cond) O O 2C-C-OH + 2Na2(C-C-O-)Na+ + H2 2CH3CO2H+2Na2(CH3CO2-)Na+ +H2 Sodium ethanoate
Examples To name acid salts: • Name Metal ion • Drop –ic acid and add –ate Ex: React butyric acid with magnesium (str,cond,name)
Acid Salt Formation – 2nd Way! 2. Carboxylic acid + metal hydroxide acid salt + water RCO2H + MOH(RCO2-)M+ + H2O React butanoic acid with magnesium hydroxide (str,cond, name prod)
Example React propanoic acid with aluminum hydroxide (str, cond, name product)
Other Reactions of Carboxylic Acids Convert carboxylic acids to “functional derivatives” Functional Derivative – an organic compound containing the acyl group O Acyl group = RCO = R-C-
Conversion to acid chlorides O O 4. R-C-OH + SOCl2R-C-Cl + SO2 + HCl RCO2H + SOCl2 RCOCl + SO2 + HCl Carboxylic acid + thionyl chloride acid chloride + sulfur dioxide + hydrogen chloride
Examples React hexanoixc acid with thionyl chloride (s,c,np) O O C-C-C-C-C-C-OH + SOCl2C-C-C-C-C-C-Cl + SO2+HCl CH3(CH2)4CO2H + SOCl2 CH3(CH2)4COCl + SO2+HCl To name acid chlorides: • Drop –ic acid • Add –yl chloride
Conversion to amides 5. Converting carbox. Acids to amides O H O H R-C-OH + H-N-H R-C-N-H + H2O Carboxylic acid + ammonia amide + water
Example Rctpentanoic acid w/ammonia (s,c,np) O H O H C-C-C-C-C-OH + H-N-H C-C-C-C-C-N-H + H2O CH3(CH2)3COH + NH3 CH3(CH2)3CONH2 + H2O To Name Amides: pentanimide • Drop –e from corresponding alkane • Add –amide
Conversions to Esters Fischer Esterification O O R-C-OH + R’-OH H2SO4 R-C-O-R’ + H2O RCO2H + R’OH H2SO4 RCO2R’ + H2O Carboxylic acid + alcoholester + water
Example React propanoic acid with butanol (s,c,np) O O C-C-C-OH + C-C-C-C-OH H2SO4 C-C-C-O-C-C-C-C + H2O CH3CH2CO2H + CH3(CH2)2CH2OHH2SO4 CH3CH2CO2(CH2)3CH3+ H2O butyl propanoate
Naming Esters To Name Esters: • Name R’ side as chain (from alcohol) • Drop – ic acid add – ate EX: form Propyl Butanoate (s,c,nr)