Chemical characteristic properties
This resource covers the chemical characteristics of substances and their reactions to various indicators. It examines how litmus paper reveals whether a substance is acidic or basic. The interaction with cobalt chloride paper and limewater is explained, showcasing color changes and reactions with carbon dioxide. Furthermore, it highlights the behavior of glowing and burning splints in the presence of oxygen and hydrogen, including historical context from the Hindenburg incident. Ideal for students looking to understand chemical properties and reactions.

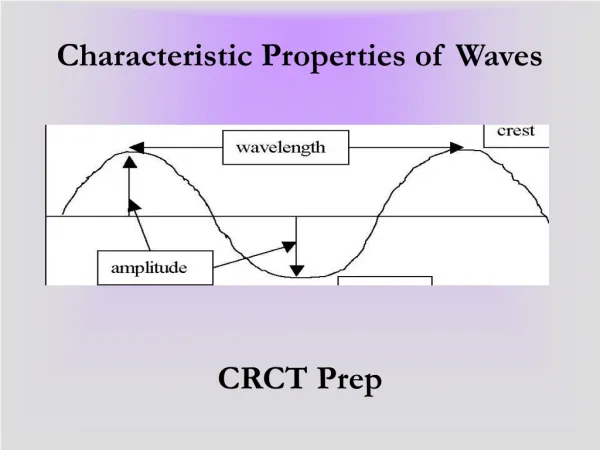
Chemical characteristic properties
E N D
Presentation Transcript
Reaction to litmus paper • Can identify if a substance is acidic or basic. Acid Base
Reaction to cobalt chloride paper • Turns pink in the presence of water. no water with water
Reaction to limewater • Turns Cloudy in the presence of CO2
Reaction to glowing splint • Splint reignites in the presence of oxygen A pair of bellows uses the same principal to reignite a fire
Reaction to burning splint • An explosion will occur in the presence of hydrogen. Hindenburg in 1937 http://www.youtube.com/watch?v=1PLH4H7Mjl8
Reaction to an open flame The color of the flame may indicate the presence of certain substances. Ba Na K http://www.youtube.com/watch?v=PVj8aQwuUMo
Homework • Workbook pages 17 and 18 (# 1 to 6)