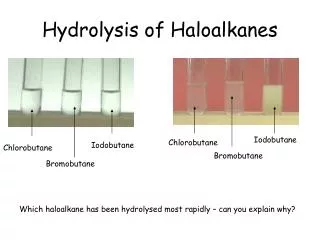
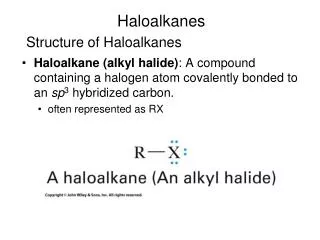CHAPTER 7 Haloalkanes
CHAPTER 7 Haloalkanes. Haloalkane (alkyl halide): a compound containing a halogen covalently bonded to an sp 3 hybridized carbon; given the symbol RX. Haloalkanes. An alkane in which one or more H atoms is replaced with a halogen (F, Cl , Br, or I) CH 3 Br 1-bromomethane

CHAPTER 7 Haloalkanes
E N D
Presentation Transcript
CHAPTER 7 Haloalkanes
Haloalkane (alkyl halide):a compound containing a halogen covalently bonded to an sp3 hybridized carbon; given the symbol RX
Haloalkanes An alkane in which one or more H atoms is replaced with a halogen (F, Cl, Br, or I) CH3Br 1-bromomethane Br (methyl bromide) CH3CH2CHCH3 2-bromobutane Cl chlorocyclobutane Timberlake LecturePLUS 1999
Nomenclature - IUPAC • locate the parent alkane • number the parent chain to give the substituent encountered first the lower number • show halogen substituents by the prefixes fluoro-, chloro-, bromo-, and iodo- and list them in alphabetical order with other substituents • locate each halogen on the parent chain
Nomenclature • examples • Common names: name the alkyl group followed by the name of the halide
Nomenclature • several polyhaloalkanes are common solvents and are generally referred to by their common or trivial names • hydrocarbons in which all hydrogens are replaced by halogens are commonly named as perhaloalkanes or perhaloalkenes
Name the following: Timberlake LecturePLUS 1999
Name the following: bromocyclopentane 1,3-dichlorocyclohexane Timberlake LecturePLUS 1999
SN2 – Substitution Nucleophilic, Bimolecular • This is called a concerted reaction • Meaning that the bond breaking and the bond forming occur simultaneously • Is classified as bimolecular • Because both the haloalkane and the nucleophile are involved in the rate determining step. • S = substitution • N = nucleophilic • 2 = bimolecular (two species are involved in the rate-determining step)
Recall • Nucleophile(nucleus loving): An electron rich species that seeks a region of low electron density (Nu). • Electrophile (electron loving): A low electron-density species that seeks a region of high electron density.
SN2 – Substitution Nucleophilic, Bimolecular The nucleophile attacks the reactive center from the side opposite the leaving group; in other words it involves a backside attack by the nucleophile.
SN2 • both reactants are involved in the transition state of the rate-determining step • the nucleophile attacks the reactive center from the side opposite the leaving group
Press the slide show button to see the animation. Press ESC to finish. ENERGY PROFILE SN2 ANIMATION R .. C Cl: .. H CH3
.. :Br: .. ENERGY PROFILE SN2 ANIMATION R .. C Cl: .. H CH3
.. :Br: .. ENERGY PROFILE SN2 ANIMATION R .. C Cl: .. H CH3
.. :Br: .. ENERGY PROFILE SN2 ANIMATION R .. C Cl: .. H CH3
.. :Br: .. ENERGY PROFILE SN2 ANIMATION R .. C Cl: .. H CH3
.. .. :Br Cl: .. .. ENERGY PROFILE SN2 ANIMATION R Transition State d- d- C H CH3 Activated Complex
.. .. :Br :Cl: .. .. ENERGY PROFILE SN2 ANIMATION R C H CH3
.. .. :Br :Cl: .. .. ENERGY PROFILE SN2 ANIMATION R C H CH3
.. .. :Br :Cl: .. .. ENERGY PROFILE SN2 ANIMATION R C H CH3
.. :Br .. ENERGY PROFILE SN2 ANIMATION R C H CH3
SN1 – Substitution Nucleophilic, Unimolecular • S = substitution • N = nucleophilic • 1 = unimolecular (only one species is involved in the rate-determining step)
SN1 – Substitution Nucleophilic, Unimolecular • In this reaction the bond breaking between carbon and the leaving group is entirely completed before bond forming with the nucleophile begins • This is classified as unimolecular • Only the haloalkane is involved in the rate-determining step • In other words, only the haloalkane contributes to the rate law governing the rate determining step
SN1 • Step 1: ionization of the C-X bond gives a carbocation intermediate
SN1 • Step 2: reaction of the carbocation (an electrophile, low electron density) with methanol (a nucleophile, high electron density) gives an oxonium ion • Step 3: proton transfer completes the reaction
SN1 • For an SN1 reaction at a stereocenter, the product is a racemic mixture
SN1 • the nucleophile attacks with equal probability from either face of the planar carbocation intermediate
SN2 and SN1 • Are competing constantly, what determines what mechanism is a reaction going to prefer? 1.The structure of the nucleophile 2.The structure of the haloalkane 3.The leaving group 4.The solvent
1. The structure of the nuceophile • Refer to table 7.2 page 228 from your book to see the types of nucleophiles we deal with most commonly in this semester. • Nucleophilicity: a kinetic property measured by the rate at which a Nu attacks
Nucleophilicity • Table 7.2
2. Structure of the Haloalkane • SN1 reactions • governed by electronic factors, namely the relative stabilities of carbocation intermediates • relative rates: 3° > 2° > 1° > methyl • SN2 reactions • governed by steric factors, namely the relative ease of approach of the nucleophile to the site of reaction • relative rates: methyl > 1° > 2° > 3°
SN1 • SN1 will be favored if a tertirarycarbocation is involved, sometimes if a secondary carbocation is involved • SN1 will never be favored if a primary cabocation or methyl are involved
SN2 • The less crowded site will always favor the SN2 mechanism • Will be favored if it involves a primary carbocation and methyl • Sometimes will be favored if a secondary carbocation is involved
3.Leaving group • Chlorine ion, bromine ion and Iodine ion make good leaving groups because of their size and Electronegativity help to stabilize the resulting negative charge • The ability of a group to function as a leaving group is related to how stable is as an anion • The most stable anion and the best leaving groups are the conjugate bases of strong acids!!!
4. The Solvent • Protic solvent: a solvent that contains an -OH group • these solvents favor SN1 reactions; the greater the polarity of the solvent, the easier it is to form carbocations in it
4. The Solvent • Aproticsolvent:does not contain an -OH group • it is more difficult to form carbocations in aprotic solvents • aprotic solvents favor SN2 reactions
Elimination reactions • Dehydrohalogenation • These reaction require forcing conditions like a strong base and heat. • (Hydroxide ion or ethoxide ion) • Halogen is removed from one carbon of a haloalkane • And the hydrogen from the adjacent carbon • To form a double bond • (an alkene)
b-Elimination • Zaitsev rule: the major product of a -elimination is the more stable (the more highly substituted) alkene
E1 and E2 mechanisms • There are both examples of beta-elimination reactions • The difference is the timing of the bond-breaking and the bond-forming steps. • E1 stands for elimination and 1 for unimolecular • E2 stands for elimination and 2 for bimolecular
E1 • The breaking for the halogen carbon bond has to be completely broken before any reaction occurs with the base • This is the slow determining step (the breaking of the halogen carbon bond)
E1 Mechanism • Step 1: The breaking for the halogen carbon bond gives a carbocation intermediate • Step 2: proton transfer from the carbocation intermediate to a base (in this case, the solvent) gives the alkene
E2 • The base removes a beta hydrogen at the same time that carbon halogen bond is broken • The rate of the reaction will depend both on the haloalkane and the base • The stronger the base the more likely it is that the E2 mechanism will be in operation
E2 Mechanism • A one-step mechanism; all bond-breaking and bond-forming steps are concerted
Table 7.6 • E2 is favored if you are dealing with a primary haloalkane • E2 is favored for secondary haloalkane if you have a really strong base • E1 is favored for secondary haloalkane if you have weak bases • E1 is favored for tertiary haloalkanes.