Solubility curves
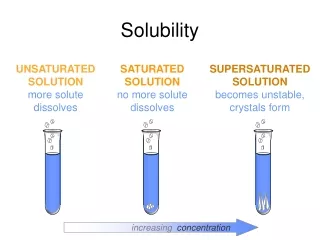
Solubility curves. Class data. Write down the data & answer questions on bottom of the sheet. Results of lab: solubility of KClO 3. KNO 3. 3a i) 7 . Unsaturated. ii) 14. Supersaturated. 4 i). iii) 24 - 25. Saturated. 3b i) 30 °C. 4 ii). ii) 3 °C. 4iii). iii) 48°C.

Solubility curves
E N D
Presentation Transcript
Class data Write down the data & answer questions on bottom of the sheet
Results of lab: solubility of KClO3 KNO3 3a i) 7 Unsaturated ii) 14 Supersaturated 4 i) iii) 24 - 25 Saturated 3b i) 30°C 4 ii) ii) 3°C 4iii) iii) 48°C 4 i) saturated ii) unsaturated iii) unsaturated
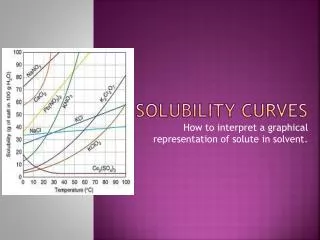
Saturation and solubility curves In this experiment we expressed concentration in units of g solute / g H2O Putting concentrations in g solute / g solution would be tedious and impractical See Fig 2 on pg. 316 Notice that there are solubility curves for many different substances Q - Why do temperatures range from 0-100°C A - Water is a liquid at these temperatures Solubility curves tell us how much solid will dissolve at a given temperature They also allow certain calculations…
Calculations using solubility curves Using fig 2 (pg. 316) 5. 100 g H2O is saturated with KClO3 at 100°C. How much KClO3 will come out of solution if the temperature is decreased to 48°C? How much would precipitate under the same circumstances if 350 g water were used 6. 57 g - 18 g = 39 g (57 g - 18 g) x 3.5 = 137 g (85 g - 21 g) x 1.5 = (64 g) x 1.5 = 96 g For more lessons, visit www.chalkbored.com